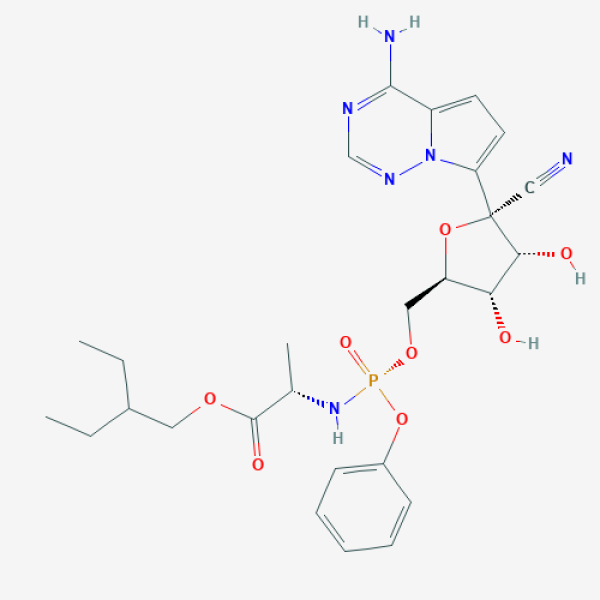
|
NCT04323761
|
Expanded Access Treatment Protocol: Remdesivir (RDV; GS-5734) for the Treatment of SARS-CoV2 (CoV) Infection (COVID-19) |
Approved for marketing |
|
Jan/01/1970 |
Jan/01/1970 |
- Alternative id - GS-US-540-5821|2020-001453-49
- Interventions - Drug: Remdesivir
- Study type - Expanded Access:Treatment IND/Protocol
- Study results - No Results Available
- Locations - University of Alabama- Birmingham, Birmingham, Alabama, United States|Banner- University Medical Center Phoenix, Phoenix, Arizona, United States|Community Regional Medical Centers (CRMC), Fresno, California, United States|St. Jude Medical Center, Fullerton, California, United States|Scripps Memorial Hospital La Jolla, La Jolla, California, United States|Long Beach Memorial Medical Center, Long Beach, California, United States|Huntington Hospital, Pasadena, California, United States|Scripps Mercy Hospital, San Diego, California, United States|California Pacific Medical Center, San Francisco, California, United States|University of California, Medical Center (Parnassus Campus), San Francisco, California, United States|Regional Medical Center, San Jose, California, United States|San Mateo Medical Center, San Mateo, California, United States|Santa Rosa Memorial Hospital, Santa Rosa, California, United States|Los Robles Regional Medical Center, Thousand Oaks, California, United States|PIH Health Whittier Hospital, Whittier, California, United States|The Medical Center Of Aurora, Aurora, Colorado, United States|Rose Medical Center, Denver, Colorado, United States|Swedish Medical Center, Englewood, Colorado, United States|Greenwich Hospital, Greenwich, Connecticut, United States|Middlesex Health, Middletown, Connecticut, United States|The Hospital of Central Connecticut, New Britain, Connecticut, United States|The Stamford Health Medical Group- Pulmonary Associates, Stamford, Connecticut, United States|George Washington University Hospital, Washington, District of Columbia, United States|Aventura Hospital and Medical Center, Aventura, Florida, United States|Lawnwood Regional Medical Center, Fort Pierce, Florida, United States|Memorial Regional Hospital, Hollywood, Florida, United States|Mayo Clinic in Florida, Jacksonville, Florida, United States|Mount Sinai Medical Center, Miami Beach, Florida, United States|AdventHealth Orlando, Investigational Drug Services, Orlando, Florida, United States|Sarasota Memorial Hospital, Sarasota, Florida, United States|Piedmont Atlanta Hospital, Atlanta, Georgia, United States|Wellstar Atlanta Medical Center, Atlanta, Georgia, United States|Piedmont Columbus Regional-Midtown Hospital, Columbus, Georgia, United States|Grady Health System- Grady Memorial Hospital, Decatur, Georgia, United States|Piedmont Fayette Hospital, Fayetteville, Georgia, United States|Memorial Health University Medical Center, Savannah, Georgia, United States|John D. Archbold Memorial Hospital, Thomasville, Georgia, United States|Swedish Hospital, Chicago, Illinois, United States|Mercy Hospital & Medical Center, Chicago, Illinois, United States|Northwestern Lake Forest Hospital, Lake Forest, Illinois, United States|Loyola University Medical Center, Maywood, Illinois, United States|Northwestern Medicine Central DuPage Hospital, Winfield, Illinois, United States|St. Vincent Hospital and Health Care Center, Inc., Carmel, Indiana, United States|Richard L Roudebush Indianapolis VA, Indianapolis, Indiana, United States|Riverview Health, Noblesville, Indiana, United States|Our Lady of the Lakes Regional Medical Center, Baton Rouge, Louisiana, United States|Touro Infirmary, New Orleans, Louisiana, United States|Ochsner Clinic Foundation, New Orleans, Louisiana, United States|Slidell Memorial Hospital, Slidell, Louisiana, United States|University of Maryland, Baltimore, Maryland, United States|Mercy Medical Center, Baltimore, Maryland, United States|CalvertHealth Medical Center, Prince Frederick, Maryland, United States|University of Maryland St. Joseph Medical Center, Towson, Maryland, United States|Steward St. Elizabeth's Medical Center of Boston, Inc., Boston, Massachusetts, United States|Cambridge Health Alliance, Cambridge, Massachusetts, United States|Charlton Memorial Hospital, Fall River, Massachusetts, United States|Lawrence General Hospital, Lawrence, Massachusetts, United States|Lowell General Hospital, Lowell, Massachusetts, United States|South Shore Hospital, South Weymouth, Massachusetts, United States|UMass Memorial Medical Center, Worcester, Massachusetts, United States|St. Joseph Mercy Hospital, Ann Arbor, Michigan, United States|Huron Valley-Sinai Hospital, Commerce, Michigan, United States|Wayne Statue University/ Detroit Receiving Hospital, Detroit, Michigan, United States|Henry Ford Health System, Detroit, Michigan, United States|McLaren Health Care Corporation, Flint, Michigan, United States|Bronson Methodist Hospital, Kalamazoo, Michigan, United States|University of Mississippi Medical Center, Jackson, Mississippi, United States|Washington University School of Medicine, Saint Louis, Missouri, United States|CarePoint Health Bayonne Medical Center, Bayonne, New Jersey, United States|Clara Maass Medical Center, Belleville, New Jersey, United States|Cooper University Hospital, Camden, New Jersey, United States|Hackensack Meridian Health - JFK Medical Center, Edison, New Jersey, United States|Trinitas Regional Medical Center, Elizabeth, New Jersey, United States|Englewood Health, Englewood, New Jersey, United States|Hunterdon Medical Center, Flemington, New Jersey, United States|CentraState Medical Center, Freehold, New Jersey, United States|Robert Wood Johnson University Hospital Hamilton, Hamilton, New Jersey, United States|Jersey City Medical Center, Jersey City, New Jersey, United States|Christ Hospital- Carepoint Health, Jersey City, New Jersey, United States|Monmouth Medical Center, Long Branch, New Jersey, United States|Atlantic Health System/ Morristown Medical Center, Morristown, New Jersey, United States|Jersey Shore University Medical Center, Neptune, New Jersey, United States|Saint Peter's University Hospital, New Brunswick, New Jersey, United States|Saint Michael's Medical Center, Newark, New Jersey, United States|University Hospital, Newark, New Jersey, United States|Ocean Medical Center, Oakhurst, New Jersey, United States|Bayshore Medical Center, Red Bank, New Jersey, United States|The Valley Hospital, Ridgewood, New Jersey, United States|Atlantic Health System/Overlook Medical Center, Summit, New Jersey, United States|Holy Name Medical Center, Teaneck, New Jersey, United States|Capital Health, Trenton, New Jersey, United States|Virtua Health Voorhees Campus, Voorhees, New Jersey, United States|St. Peter's Hospital, Albany, New York, United States|Mount Sinai Queens, Astoria, New York, United States|Good Samaritan Hospital Medical Center, Bay Shore, New York, United States|Southside Hospital, Bay Shore, New York, United States|Lincoln Medical Center, Bronx, New York, United States|St Barnabus Hospital, Bronx, New York, United States|Montefiore Medical Center, Weiler Campus, Bronx, New York, United States|Montefiore Health System Moses Medical Center, Bronx, New York, United States|The Brooklyn Hospital Center, Brooklyn, New York, United States|Kingsbrook Jewish Medical Center, Brooklyn, New York, United States|University Hospital Brooklyn (SUNY Downstate), Brooklyn, New York, United States|Woodhull Medical Center, Brooklyn, New York, United States|Kings County Hospital Center, Brooklyn, New York, United States|New York Harbor VA Medical Center, Brooklyn, New York, United States|Brookdale University Hospital and Medical Center, Brooklyn, New York, United States|Interfaith Medical Center, Brooklyn, New York, United States|NewYork-Presbyterian Brooklyn Methodist Hospital, Brooklyn, New York, United States|Maimonides Medical Center, Brooklyn, New York, United States|NYU Langone Hospital - Brooklyn, Brooklyn, New York, United States|Mount Sinai Brooklyn, Brooklyn, New York, United States|New York City Health and Hospitals - Coney Island, Brooklyn, New York, United States|Kaleida Health System, Buffalo, New York, United States|Nassau University Medical Center, East Meadow, New York, United States|New York Presbyterian- Queens, Flushing, New York, United States|Glens Falls Hospital, Glens Falls, New York, United States|Queens Hospital Center, Jamaica, New York, United States|NYU Langone Health, New York, New York, United States|Hospital for Special Surgery, New York, New York, United States|New York City Health and New York City Health and Hospitals - Metropolitan- Metropolitan, New York, New York, United States|NewYork-Presyterian/Columbia University Irving Medical Center- Milstein Hospital, New York, New York, United States|New York City Health + Hospitals / Harlem, New York, New York, United States|New York Presbyterian Lower Manhattan Hospital, New York, New York, United States|Memorial Sloan-Kettering Cancer Center, New York, New York, United States|Mount Sinai South Nassau, Oceanside, New York, United States|Vassar Brothers Medical Center, Poughkeepsie, New York, United States|Rochester General Hospital, Rochester, New York, United States|St. Francis Hospital, Roslyn, New York, United States|Ellis Hospital, Schenectady, New York, United States|Staten Island University Hospital- Northwell Health (North Campus), Staten Island, New York, United States|Stony Brook University Hospital/ Stony Brook Children's, Stony Brook, New York, United States|Samaritan Hospital, Troy, New York, United States|UNC Hospitals, Chapel Hill, North Carolina, United States|Atrium Health- Carolinas Medical Center, Charlotte, North Carolina, United States|MetroHealth Medical Center, Cleveland, Ohio, United States|The Ohio State University College of Medicine, Columbus, Ohio, United States|Kettering Medical Center, Kettering, Ohio, United States|Saint Francis Hospital, Tulsa, Oklahoma, United States|St. Luke's Hospital - Bethlehem Campus, Bethlehem, Pennsylvania, United States|Mercy Catholic Medical Center - Fitzgerald Campus, Darby, Pennsylvania, United States|St Mary Medical Center, Langhorne, Pennsylvania, United States|Philadelphia VA Medical Center, Philadelphia, Pennsylvania, United States|Einstein Medical Center Philadelphia, Philadelphia, Pennsylvania, United States|Allegheny Health Network, Pittsburgh, Pennsylvania, United States|UPMC, Pittsburgh, Pennsylvania, United States|Roger Williams Medical Center, Providence, Rhode Island, United States|Kent Hospital, Warwick, Rhode Island, United States|St. Francis Hospital, Greenville, South Carolina, United States|Lexington Medical Center, West Columbia, South Carolina, United States|TriStar Centennial Medical Center, Nashville, Tennessee, United States|St. David's South Austin Medical Center, Austin, Texas, United States|Texas Health Presbyterian Hospital Dallas, Dallas, Texas, United States|Texas Department of Criminal Justice Hospital (Hospital Galveston)/University of Texas Medical Branch, Galveston, Texas, United States|HCA Houston Healthcare West, Houston, Texas, United States|Covenant Medical Center, Lubbock, Texas, United States|Methodist Mansfield Medical Center, Mansfield, Texas, United States|Methodist Healthcare System of San Antonio, Ltd. LLP, dha, Methodist Hospital, San Antonio, Texas, United States|Saint Mark's Hospital, Salt Lake City, Utah, United States|Inova Fairfax Medical Campus, Falls Church, Virginia, United States|Henrico Doctors Hospital, Richmond, Virginia, United States|Carilion Medical Center, Roanoke, Virginia, United States|Overlake Hospital Medical Center, Bellevue, Washington, United States|University of Washington Medical Center- Montlake Campus, Seattle, Washington, United States|Medical College of Wisconsin, Milwaukee, Wisconsin, United States|St. Vincent's Hospital Sydney, Darlinghurst, New South Wales, Australia|Royal Brisbane and Women's Hospital, Herston, Queensland, Australia|The Alfred Hospital, Melbourne, Victoria, Australia|The Royal Melbourne Hospital, Parkville, Victoria, Australia|Gold Coast Hospital, Southport, Australia|Landeskrankenhaus Graz West, Graz, Austria|Universitätsklinik für Innere Medizin II, Innsbruck, Austria|Kepler Universitätsklinikum, Linz, Austria|Medical University and General Hospital Vienna, Vienna, Austria|Sozialmedizinisches Zentrum Süd - Kaiser-Franz-Josef-Spital, Wien, Austria|Centre Hospitalier Universitaire Saint Pierre, Brussels, Belgium|Ghent University Hospital, Ghent, Belgium|Jessa Ziekenhuis, Hasselt, Belgium|AZ Groeninge, Kortrijk, Belgium|Universitair Ziekenhuis Leuven, Leuven, Belgium|Centre Hospitalier Universitaire de Liège, Liège, Belgium|Hôpital Civil Marie Curie, Lodelinsart, Belgium|William Osler Health System, Brampton, Ontario, Canada|The Ottowa Hospital, Ottawa, Ontario, Canada|Nicosia General Hospital, Nicosia, Cyprus|Všeobecná fakultní nemocnice v Praze, Prague, Czechia|Eticka komise pro multicentircke klinicke hodnoceni Fakultni nemocnice v Motole, Praha 5, Czechia|Nemocnice Na Bulovce, Praha 8 - Liben, Czechia|Rigshospitalet, Copenhagen, Denmark|Põhja-Eesti Regionaalhaigla, Tallinn, Estonia|Vilnius University, Tartu, Estonia|Hopital Avicenne, Bobigny, France|Centre Hospitalier Regional Universitaire Brest Hôpital de la Cavale Blanche à Brest, Brest Cedex, France|Centre Hospitalier Universitaire Gabriel-Montpied, Clermont Ferrand, France|Hôpitaux Civils de Colmar, Colmar, France|Centre Hospitalier Sud Francilien, Corbeil Essonnes, France|Hôpital Raymond Poincaré, Garches, France|Centre Hospitalier Régional d'Orléans, Orléans, France|Centre Hospitalier Universitaire de Poitiers, Poitiers, France|Hôpital Charles-Nicolle, Rouen, France|Hôpital Purpan, Toulouse, France|Saint Josef Hospital Ruhr Universitaet Bochum, Bochum, Germany|Universitätsklinikum Carl Gustav Carus Dresden, Dresden, Germany|Universitatsklinikum, Essen, Germany|Universitätsklinikum Freiburg, Freiburg, Germany|Universitätsmedizin Göttingen, Göttingen, Germany|Medizinische Hochschule Hannover, Hannover, Germany|Universitätsklinikum Heidelberg, Heidelberg, Germany|Universitätsklinikum des Saarlandes, Homburg, Germany|Caritas-Krankenhaus St. Josef, Regensburg, Germany|Klinikum Würzburg Mitte, Würzburg, Germany|University General Hospital of Patras Panagia I Voithia, Patra, Greece|General Hospital of Thessaloniki George Papanikolaou, Thessaloniki, Greece|Kenézy Gyula Kórház És Rendelőintézet, Debrecen, Hungary|Landspítali, Reykjavik, Iceland|Kerry University Hospital, Tralee, Ireland|Shaare Zedek Medical Center, Jerusalem, Israel|Hasharon Hospital, Petah Tikva, Israel|Sheba Medical Center at Tel Hashomer, Ramat Gan, Israel|Azienda Ospedaliero Universitaria Ospedali Riuniti di Ancona, Ancona, Italy|Azienda Universitaria Ospedaliera Consorziale - Policlinico di Bari, Bari, Italy|Azienda Ospedaliero Universitaria, Bologna, Italy|ASST Spedali Civili di Brescia, Brescia, Italy|Presidio Ospedaliero Garibaldi-Nesima, Catania, Italy|Azienda Ospedaliero - Universitaria Careggi, Florence, Italy|Ospedale Policlinico San Martino, Genova, Italy|Ospedale Maggiore Policlinico, Milano, Italy|Ospedale San Gerardo di Monza, Monza, Italy|Azienda Ospedaliera dei Colli, Napoli, Italy|Azienda Ospedaliero-Universitaria Pisana, Pisa, Italy|IRCCS Istituto Nazionale per le Malattie Infettive Lazzaro Spallanzani, Roma, Italy|Azienda Ospedaliero Universitaria, Turin, Italy|Hospital of Lithuanian University of Health Sciences Kauno klinikos, Kaunas, Lithuania|Vilnius University, Vilnius, Lithuania|Radboud Universitair Medisch Centrum, Nijmegen, Netherlands|HagaZiekenhuis, The Hague, Netherlands|Szpital Uniwersytecki w Krakowie, Krakow, Poland|Wojewódzki Specjalistyczny Szpital im. dr. Wł. Biegańskiego, Lodz, Poland|Wielospecjalistyczny Szpital Miejski Im Józefa Strusia Zakład Opiekuńczo Leczniczy, Poznan, Poland|Wojewódzki Szpital Specjalistyczny Megrez Sp. z o.o., Tychy, Poland|Centro Hospitalar Universitário de Lisboa Central - Hospital Curry Cabral, Lisboa, Portugal|Centro Hospitalar Universitário do Porto E.P.E., Porto, Portugal|Centro Hospitalar Universitário De São João, Porto, Portugal|Centro Hospitalar de Vila Nova de Gaia/Espinho, Vila Nova de Gaia, Portugal|Institutul National de Boli Infectioase Prof. Dr. Matei Bals, Bucharest, Romania|Spitalul Clinic de Boli Infecţioase Sfânta Parascheva Iaşi, Iasi, Romania|Clinical hospital for infectious disease, Timisoara, Romania|Univerzitná Nemocnica L. Pasteura Košice - Pracovisko Rastislavova 43, Košice, Slovakia|Univerzitetni Klinicni Center Ljubljana, Ljubljana, Slovenia|Univerzitetni Klinicni Center Maribor, Maribor, Slovenia|Hospital Universitari Germans Trias i Pujol, Badalona, Spain|Hospital General Universitario Gregorio Maranon, Madrid, Spain|Hospital Universitario Central de Asturias, Oviedo, Spain|Hospital Universitario Son Espases, Palma de Mallorca, Spain|Hospital Clinico Universitario de Salamanca, Salamanca, Spain|Hospital Universitario Donostia, San Sebastian, Spain|Hospital Virgen De La Salud, Santa Cruz de Tenerife, Spain|Hospital Virgen De La Salud, Toledo, Spain|Hospital Clinico Universitario de Valladolid, Valladolid, Spain|Hospital Universitario Miguel Servet, Zaragoza, Spain|Kantonsspital Aarau, Aarau, Switzerland|University Hospital Basel, Basel, Switzerland|Spitalzentrum Biel, Biel, Switzerland|Kantonsspital Graubünden, Chur, Switzerland|Centre Hospitalier Universitaire Vaudois Lausanne, Lausanne, Switzerland|Luzerner Kantonsspital, Luzern, Switzerland|Hôpital du Valais -Hôpital de Sion, Sion, Switzerland|University Hospital of Wales, Cardiff and Vale NHS Trust, Cardiff, United Kingdom|Barts Health NHS Trust, London, United Kingdom|Guy's and St Thomas' NHS Foundation Trust, London, United Kingdom|St. George's Hospital, London, United Kingdom|Southampton University Hospital, Southampton, United Kingdom
- Study designs -
- Enrollment -
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures -
|
|
NCT04988035
|
ACTIV-5 / Big Effect Trial (BET-C) for the Treatment of COVID-19 |
Recruiting |
Phase 2 |
Jul/21/2021 |
Dec/22/2022 |
- Alternative id - 20-0013C
- Interventions - Drug: Danicopan|Other: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - University of Alabama at Birmingham School of Medicine - Infectious Disease, Birmingham, Alabama, United States|Kern Medical Center, Bakersfield, California, United States|UCSF Fresno Center for Medical Education and Research - Clinical Research Center, Fresno, California, United States|University of California Los Angeles Medical Center - Westwood Clinic, Los Angeles, California, United States|Hoag Hospital Newport Beach, Newport Beach, California, United States|Penrose Hospital - Emergency Medicine, Colorado Springs, Colorado, United States|St. Francis Medical Center, Colorado Springs, Colorado, United States|St. Anthony Hospital, Lakewood, Colorado, United States|St. Anthony Hospital North Health Campus, Westminster, Colorado, United States|Nuvance Health Danbury Hospital - Infectious Disease, Danbury, Connecticut, United States|Yale School of Medicine - The Anlyan Center for Medical Research & Education - Immunobiology, New Haven, Connecticut, United States|Nuvance Health - Norwalk Hospital - Asthma Pulmonary and Critical Care Medicine, Norwalk, Connecticut, United States|University of Florida Health - Shands Hospital - Division of Infectious Diseases and Global Medicine, Gainesville, Florida, United States|Mayo Clinic Florida, Jacksonville, Florida, United States|Great Lakes Clinical Trials, Chicago, Illinois, United States|Carle Foundation Hospital, Urbana, Illinois, United States|Northwestern Medicine - Central DuPage Hospital - Infectious Disease, Winfield, Illinois, United States|Norton Healthcare, Louisville, Kentucky, United States|University of Louisville - Division of Infectious Diseases, Louisville, Kentucky, United States|Brigham and Women's Hospital - Infectious Diseases, Boston, Massachusetts, United States|William Beaumont Hospital - Royal Oak Campus - Infectious Disease, Royal Oak, Michigan, United States|Hennepin Healthcare Research Institute, Minneapolis, Minnesota, United States|Mayo Clinic, Rochester - Infectious Diseases, Rochester, Minnesota, United States|University of Nebraska Medical Center - Infectious Diseases, Omaha, Nebraska, United States|Jacobi Medical Center, Bronx, New York, United States|Montefiore Medical Center - Infectious Diseases, Bronx, New York, United States|The State University of New York - University at Buffalo - Department of Medicine, Buffalo, New York, United States|Mount Sinai School of Medicine - Medicine - Infectious Diseases, New York, New York, United States|Nuvance Health - Vassar Brothers Medical Center, Poughkeepsie, New York, United States|Stony Brook Medicine - Stony Brook University Hospita, Stony Brook, New York, United States|Wake Forest Baptist Health - Infectious Diseases, Winston-Salem, North Carolina, United States|St. Charles Health System - St. Charles Bend Hospital, Bend, Oregon, United States|Doylestown Hospital, Doylestown, Pennsylvania, United States|Penn State Health Milton S. Hershey Medical Center - Division of Infectious Diseases, Hershey, Pennsylvania, United States|Kent County Memorial Hospital, Warwick, Rhode Island, United States|Hendrick Health - Hendrick Medical Center, Abilene, Texas, United States|Baylor Scott & White Health - Baylor University Medical Center - North Texas Infectious Disease Consultants, Dallas, Texas, United States|University of Utah - Infectious Diseases, Salt Lake City, Utah, United States|West Virginia University - Infectious Diseases Clinic, Morgantown, West Virginia, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 200
- Age - Child, Adult, Older Adult
- Outcome measures - Clinical status on an 8-point ordinal scale.|Change from baseline of inflammation and coagulation markers|Change in alanine aminotransferase (ALT) over time|Change in aspartate transaminase (AST) over time|Change in creatinine over time|Change in hemoglobin over time|Change in international normalized ratio (INR)|Change in platelets over time|Change in total bilirubin over time|Change in white blood cell (WBC) count with differential over time|Clinical Status on an 8-point ordinal scale|Cumulative incidence of Grade 3 and 4 clinical and/or laboratory adverse events|Cumulative incidence of serious adverse events (SAE)|Discontinuation or temporary suspension of study product administration|Duration of hospitalization|Duration of Intensive Care Unit stay|Duration of mechanical ventilation or extracorporeal membrane oxygenation (ECMO) use|Duration of new mechanical ventilation or extracorporeal membrane oxygenation (ECMO) use|Duration of new non-invasive ventilation/high-flow oxygen use|Duration of non-invasive ventilation/high-flow oxygen use|Duration of supplemental oxygen use|Incidence of new mechanical ventilation or extracorporeal membrane oxygenation (ECMO) use|Incidence of new non-invasive ventilation/high-flow oxygen use|Mean change in the ordinal scale|Subject mortality|The proportion of subjects with a baseline score of 5 or 6 who are alive and without respiratory failure|The proportion of subjects with any baseline score who are alive and without respiratory failure|Time to an improvement of one category using an ordinal scale.|Time to an improvement of two categories using an ordinal scale.|Time to death|Time to recovery
|
|
NCT04583956
|
ACTIV-5 / Big Effect Trial (BET-A) for the Treatment of COVID-19 |
Completed |
Phase 2 |
Oct/14/2020 |
Sep/13/2021 |
- Alternative id - 20-0013A
- Interventions - Other: Placebo|Drug: Remdesivir|Biological: Risankizumab
- Study type - Interventional
- Study results - No Results Available
- Locations - The University of Arizona - Banner University Medical Center Tucson Campus - Tucson, Tucson, Arizona, United States|Kern Medical Center, Bakersfield, California, United States|Hoag Hospital Newport Beach, Newport Beach, California, United States|Stanford University - Stanford Hospital and Clinics - Pediatrics - Infectious Diseases, Stanford, California, United States|Penrose Hospital - Emergency Medicine, Colorado Springs, Colorado, United States|St. Francis Medical Center, Colorado Springs, Colorado, United States|St. Anthony Hospital, Lakewood, Colorado, United States|St. Anthony Hospital North Health Campus, Westminster, Colorado, United States|Nuvance Health Danbury Hospital - Infectious Disease, Danbury, Connecticut, United States|Yale School of Medicine - The Anlyan Center for Medical Research & Education - Immunobiology, New Haven, Connecticut, United States|Nuvance Health - Norwalk Hospital - Asthma Pulmonary and Critical Care Medicine, Norwalk, Connecticut, United States|Grady Memorial Hospital, Atlanta, Georgia, United States|Emory Vaccine Center - The Hope Clinic, Decatur, Georgia, United States|Cook County Health and Hospitals System - Ruth M Rothstein CORE Center, Chicago, Illinois, United States|Rush University Medical Center, Chicago, Illinois, United States|Brigham and Women's Hospital - Infectious Diseases, Boston, Massachusetts, United States|Boston Medical Center - Center for Infectious Diseases - Shapiro Center, Boston, Massachusetts, United States|Hennepin Healthcare Research Institute, Minneapolis, Minnesota, United States|Englewood Hospital, Englewood, New Jersey, United States|Jacobi Medical Center, Bronx, New York, United States|The State University of New York - University at Buffalo - Department of Medicine, Buffalo, New York, United States|Mount Sinai School of Medicine - Medicine - Infectious Diseases, New York, New York, United States|Nuvance Health - Vassar Brothers Medical Center, Poughkeepsie, New York, United States|Wake Forest Baptist Health - Infectious Diseases, Winston-Salem, North Carolina, United States|University of Toledo Medical Center - Ruppert Clinic, Toledo, Ohio, United States|Doylestown Hospital, Doylestown, Pennsylvania, United States|Kent County Memorial Hospital, Warwick, Rhode Island, United States|Monument Health - Clinical Research, Rapid City, South Dakota, United States|Hendrick Health - Hendrick Medical Center, Abilene, Texas, United States|Baptist Hospitals of Southeast Texas Site, Beaumont, Texas, United States|West Virginia University - Infectious Diseases Clinic, Morgantown, West Virginia, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 172
- Age - Child, Adult, Older Adult
- Outcome measures - Clinical status on an 8-point ordinal scale.|Change from baseline of inflammation and coagulation markers|Change in alanine aminotransferase (ALT) over time|Change in aspartate transaminase (AST) over time|Change in creatinine over time|Change in hemoglobin over time|Change in international normalized ratio (INR) over time|Change in platelets over time|Change in total bilirubin over time|Change in white blood cell (WBC) count with differential over time|Cumulative incidence of Grade 3 and 4 clinical and/or laboratory adverse events (AEs)|Cumulative incidence of serious adverse events (SAEs)|Discontinuation or temporary suspension of study product administration|Duration of hospitalization|Duration of new mechanical ventilation or extracorporeal membrane oxygenation (ECMO) use|Duration of new non-invasive ventilation or high flow oxygen use during the study|Duration of non-invasive ventilation/high flow oxygen use|Incidence of new mechanical ventilation or extracorporeal membrane oxygenation (ECMO) use|Incidence of new non-invasive ventilation or high flow oxygen use|Mean change in the ordinal scale|Proportion of subjects alive and without respiratory failure|Subject mortality|Supplemental oxygen use|Time to an improvement of one category using an ordinal scale|Time to an improvement of two categories using an ordinal scale|Time to death|Time to recovery|Ventilator/ extracorporeal membrane oxygenation (ECMO) use
|
|
NCT04583969
|
ACTIV-5 / Big Effect Trial (BET-B) for the Treatment of COVID-19 |
Active, not recruiting |
Phase 2 |
Oct/19/2020 |
Oct/29/2022 |
- Alternative id - 20-0013B
- Interventions - Biological: Lenzilumab|Other: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - University of Alabama at Birmingham School of Medicine - Infectious Disease, Birmingham, Alabama, United States|The University of Arizona - Banner University Medical Center Tucson Campus - Tucson, Tucson, Arizona, United States|University of Arkansas for Medical Sciences, Little Rock, Arkansas, United States|Kern Medical Center, Bakersfield, California, United States|UCSF Fresno Center for Medical Education and Research - Clinical Research Center, Fresno, California, United States|University of Southern California - Infectious Diseases, Los Angeles, California, United States|Hoag Hospital Newport Beach, Newport Beach, California, United States|Stanford University - Stanford Hospital and Clinics - Pediatrics - Infectious Diseases, Stanford, California, United States|Penrose Hospital - Emergency Medicine, Colorado Springs, Colorado, United States|St. Francis Medical Center, Colorado Springs, Colorado, United States|St. Anthony Hospital, Lakewood, Colorado, United States|St. Anthony Hospital North Health Campus, Westminster, Colorado, United States|Nuvance Health Danbury Hospital - Infectious Disease, Danbury, Connecticut, United States|Yale School of Medicine - The Anlyan Center for Medical Research & Education - Immunobiology, New Haven, Connecticut, United States|Nuvance Health - Norwalk Hospital - Asthma Pulmonary and Critical Care Medicine, Norwalk, Connecticut, United States|University of Florida Health - Shands Hospital - Division of Infectious Diseases and Global Medicine, Gainesville, Florida, United States|Mayo Clinic Florida, Jacksonville, Florida, United States|Grady Memorial Hospital, Atlanta, Georgia, United States|Emory Vaccine Center - The Hope Clinic, Decatur, Georgia, United States|Cook County Health and Hospitals System - Ruth M Rothstein CORE Center, Chicago, Illinois, United States|Rush University Medical Center, Chicago, Illinois, United States|Great Lakes Clinical Trials, Chicago, Illinois, United States|Carle Foundation Hospital, Urbana, Illinois, United States|Northwestern Medicine - Central DuPage Hospital - Infectious Disease, Winfield, Illinois, United States|Norton Healthcare, Louisville, Kentucky, United States|University of Louisville - Division of Infectious Diseases, Louisville, Kentucky, United States|Brigham and Women's Hospital - Infectious Diseases, Boston, Massachusetts, United States|Boston Medical Center - Center for Infectious Diseases - Shapiro Center, Boston, Massachusetts, United States|William Beaumont Hospital - Royal Oak Campus - Infectious Disease, Royal Oak, Michigan, United States|Hennepin Healthcare Research Institute, Minneapolis, Minnesota, United States|Mayo Clinic, Rochester - Infectious Diseases, Rochester, Minnesota, United States|University of Nebraska Medical Center - Infectious Diseases, Omaha, Nebraska, United States|Englewood Hospital, Englewood, New Jersey, United States|Jacobi Medical Center, Bronx, New York, United States|Montefiore Medical Center - Infectious Diseases, Bronx, New York, United States|The State University of New York - University at Buffalo - Department of Medicine, Buffalo, New York, United States|Mount Sinai School of Medicine - Medicine - Infectious Diseases, New York, New York, United States|Nuvance Health - Vassar Brothers Medical Center, Poughkeepsie, New York, United States|Stony Brook Medicine - Stony Brook University Hospita, Stony Brook, New York, United States|Atrium Health ID Consultants & Infusion Care Specialists, Charlotte, North Carolina, United States|Wake Forest Baptist Health - Infectious Diseases, Winston-Salem, North Carolina, United States|University of Toledo Medical Center - Ruppert Clinic, Toledo, Ohio, United States|St. Charles Health System - St. Charles Bend Hospital, Bend, Oregon, United States|Doylestown Hospital, Doylestown, Pennsylvania, United States|Penn State Health Milton S. Hershey Medical Center - Division of Infectious Diseases, Hershey, Pennsylvania, United States|Kent County Memorial Hospital, Warwick, Rhode Island, United States|Monument Health - Clinical Research, Rapid City, South Dakota, United States|Hendrick Health - Hendrick Medical Center, Abilene, Texas, United States|Baptist Hospitals of Southeast Texas Site, Beaumont, Texas, United States|Baylor Scott & White Health - Baylor University Medical Center - North Texas Infectious Disease Consultants, Dallas, Texas, United States|Methodist Hospital - Houston, Houston, Texas, United States|University of Texas Health Science Center at San Antonio - Infectious Diseases, San Antonio, Texas, United States|University of Utah - Infectious Diseases, Salt Lake City, Utah, United States|West Virginia University - Infectious Diseases Clinic, Morgantown, West Virginia, United States|Seoul National University Bundang Hospital - Division of Infectious Diseases, Bundang-gu Seongnam-si, Gyeonggi-do, Korea, Republic of|Seoul National University Hospital, Seoul, Jongno-gu, Korea, Republic of
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 473
- Age - Child, Adult, Older Adult
- Outcome measures - Occurrence of mechanical ventilation or death at any point through Day 29 in subjects with ordinal scores of 5 or 6 at baseline|Change from baseline of inflammation and coagulation markers|Change in alanine aminotransferase (ALT) over time|Change in aspartate transaminase (AST) over time|Change in creatinine over time|Change in hemoglobin over time|Change in international normalized ratio (INR) over time|Change in platelets over time|Change in total bilirubin over time|Change in white blood cell (WBC) count with differential over time|Cumulative incidence of Grade 3 and 4 clinical and/or laboratory adverse events (AEs)|Cumulative incidence of serious adverse events (SAEs)|Duration of hospitalization|Duration of new mechanical ventilation or extracorporeal membrane oxygenation (ECMO) use|Duration of new non-invasive ventilation or high flow oxygen use during the study|Duration of non-invasive ventilation/high flow oxygen use|Incidence of discontinuation or temporary suspension of study product administration|Incidence of new non-invasive ventilation or high flow oxygen use|Incidence of ventilator/ extracorporeal membrane oxygenation (ECMO) use|Mean change in the ordinal scale|Subject Mortality|Supplemental oxygen use|Survival without mechanical ventilation through Day 29 in subjects with ordinal scores 5 or 6 at baseline.|Survival without mechanical ventilation through Day 29.|The proportion of alive and without respiratory failure|Time to an improvement of one category using an ordinal scale|Time to an improvement of two categories using an ordinal scale|Time to death|Time to Sustained Recovery in subjects with a baseline ordinal score of 5 or 6, CRP<150mg/L and age <85 years.|Time to sustained recovery of subjects with any baseline score|Ventilator/ extracorporeal membrane oxygenation (ECMO) use
|
|
NCT04280705
|
Adaptive COVID-19 Treatment Trial (ACTT) |
Completed |
Phase 3 |
Feb/21/2020 |
May/21/2020 |
- Alternative id - 20-0006
- Interventions - Other: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - Has Results
- Locations - University of Alabama at Birmingham School of Medicine - Infectious Disease, Birmingham, Alabama, United States|University of California San Diego Health - Jacobs Medical Center, La Jolla, California, United States|University of California Los Angeles Medical Center - Westwood Clinic, Los Angeles, California, United States|University of California Irvine Medical Center - Infectious Disease, Orange, California, United States|VA Palo Alto Health Care System - Infectious Diseases, Palo Alto, California, United States|University of California Davis Medical Center - Internal Medicine - Infectious Disease, Sacramento, California, United States|Naval Medical Center San Diego - Infectious Disease Clinic, San Diego, California, United States|University of California San Francisco - Zuckerberg San Francisco General Hospital - Division of Human Immunodeficiency Virus, Infectious Disease, and Global Medicine, San Francisco, California, United States|Stanford University - Stanford Hospital and Clinics - Pediatrics - Infectious Diseases, Stanford, California, United States|Cedars Sinai Medical Center, West Hollywood, California, United States|Denver Health Division of Hospital Medicine - Main Campus, Denver, Colorado, United States|Emory Vaccine Center - The Hope Clinic, Decatur, Georgia, United States|Northwestern Hospital - Infectious Disease, Chicago, Illinois, United States|University of Illinois at Chicago College of Medicine - Division of Infectious Diseases, Chicago, Illinois, United States|Southeast Louisiana Veterans Health Care System - Section of Infectious Diseases, New Orleans, Louisiana, United States|University of Maryland School of Medicine - Center for Vaccine Development - Baltimore, Baltimore, Maryland, United States|Johns Hopkins Hospital - Medicine - Infectious Diseases, Baltimore, Maryland, United States|Walter Reed National Military Medical Center, Bethesda, Maryland, United States|National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Clinical Research Section, Bethesda, Maryland, United States|Massachusetts General Hospital - Infectious Diseases, Boston, Massachusetts, United States|University of Massachusetts Medical School - Infectious Diseases and Immunology, Worcester, Massachusetts, United States|University of Minnesota Medical Center, Fairview - Infectious Diseases and International Medicine, Minneapolis, Minnesota, United States|Saint Louis University - Center for Vaccine Development, Saint Louis, Missouri, United States|University of Nebraska Medical Center - Infectious Diseases, Omaha, Nebraska, United States|Montefiore Medical Center - Infectious Diseases, Bronx, New York, United States|New York University School of Medicine - Langone Medical Center - Microbiology - Parasitology, New York, New York, United States|University of Rochester Medical Center - Vaccine Research Unit, Rochester, New York, United States|Duke Human Vaccine Institute - Duke Vaccine and Trials Unit, Durham, North Carolina, United States|Penn State Health Milton S. Hershey Medical Center - Division of Infectious Diseases, Hershey, Pennsylvania, United States|Hospital of the University of Pennsylvania - Infectious Diseases, Philadelphia, Pennsylvania, United States|Vanderbilt University Medical Center - Infectious Diseases, Nashville, Tennessee, United States|Brooke Army Medical Center, Fort Sam Houston, Texas, United States|University of Texas Medical Branch - Division of Infectious Disease, Galveston, Texas, United States|Baylor College of Medicine - Molecular Virology and Microbiology, Houston, Texas, United States|University of Texas Health Science Center at San Antonio - Infectious Diseases, San Antonio, Texas, United States|University of Virginia - Acute Care Surgery, Charlottesville, Virginia, United States|Naval Medical Center Portsmouth - Infectious Disease Division, Portsmouth, Virginia, United States|EvergreenHealth Infectious Disease Service, Kirkland, Washington, United States|The University of Washington - Virology Research Clinic, Seattle, Washington, United States|Providence Sacred Heart Medical Center, Spokane, Washington, United States|Madigan Army Medical Center - Infectious Disease Clinic, Tacoma, Washington, United States|University of Copenhagen - Centre of Excellence for Health, Immunity and Infections (CHIP) - Department of Infectious Diseases, Copenhagen, Denmark|Universitatsklinikum Bonn, Medizinische Klinik I - Bereich Infektiologie/HIV der Medizinischen Klinik, Bonn, Nordrhein-Westfalen, Germany|Universitatsklinikum Koeln Klinik I fur Innere Medizin Klinisches Studienzentrum fur Infektiologie I, Cologne, Germany|Universitätsklinikum Frankfurt -Medizinische Klinik II - Infektiologie, Frankfurt, Germany|AHEPA University Hospital - 1st Department of Internal Medicine, Thessaloniki, Central Macedonia, Greece|Medical School of Athens University - Evangelismos Hospital - Department of Critical Care and Pulmonary Services, Athens, Greece|National Center for Global Health and Medicine Hospital - Disease Control and Prevention Center, Tokyo, Japan|Seoul National University Bundang Hospital - Division of Infectious Diseases, Bundang-gu Seongnam-si, Gyeonggi-do, Korea, Republic of|Seoul National University Hospital, Seoul, Jongno-gu, Korea, Republic of|Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán - Departamento de Infectologia, Mexico City, Mexico|Instituto Nacional de Enfermedades Respiratorias (INER) - Ismael Cosío Villegas, Mexico City, Mexico|National Centre for Infectious Diseases, Singapore, Singapore|Hospital Clinic Barcelona, Servicio de Salud Internacional, Barcelona, Cataluña, Spain|Hospital Germans Trias i Pujol - Servei Malalties Infeccioses, Barcelona, Cataluña, Spain|Royal Sussex County Hospital - Department of Intensive Care Medicine, East Sussex, Brighton, United Kingdom|Saint Thomas' Hospital - Directorate of Infection, London, London, City Of, United Kingdom|Royal Victoria Infirmary - Department of Infectious Diseases, Level 6, Ward 19, Newcastle Upon Tyne, United Kingdom|St. James's University Hospital - Infectious Diseases, Leeds, West Yorkshire, United Kingdom|John Radcliffe Hospital, Headington, Oxford, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 1062
- Age - 18 Years to 99 Years (Adult, Older Adult)
- Outcome measures - Time to Recovery|Time to Recovery by Race|Time to Recovery by Ethnicity|Time to Recovery by Sex|Change From Baseline in Alanine Transaminase (ALT)|Change From Baseline in Aspartate Transaminase (AST)|Change From Baseline in Creatinine|Change From Baseline in Glucose|Change From Baseline in Hemoglobin|Change From Baseline in Platelets|Change From Baseline in Prothrombin Time (PT)|Change From Baseline in Total Bilirubin|Change From Baseline in White Blood Cell Count (WBC)|Change From Baseline in Neutrophils|Change From Baseline in Lymphocytes|Change From Baseline in Monocytes|Change From Baseline in Basophils|Change From Baseline in Eosinophils|Change in National Early Warning Score (NEWS) From Baseline|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 1|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 3|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 5|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 8|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 11|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 15|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 22|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 29|Percentage of Participants Reporting Grade 3 and 4 Clinical and/or Laboratory Adverse Events (AEs)|Percentage of Participants Reporting Serious Adverse Events (SAEs)|Percentage of Participants Discontinued or Temporarily Suspended From Investigational Therapeutics|Duration of Hospitalization|Duration of New Non-invasive Ventilation or High Flow Oxygen Use|Duration of New Oxygen Use|Duration of New Ventilator or Extracorporeal Membrane Oxygenation (ECMO) Use|Percentage of Participants Requiring New Non-invasive Ventilation or High-flow Oxygen Use|Percentage of Participants Requiring New Oxygen Use|Percentage of Participants Requiring New Ventilator or Extracorporeal Membrane Oxygenation (ECMO) Use|Mean Change in the Ordinal Scale|14-day Participant Mortality|29-day Participant Mortality|Time to an Improvement by at Least One Category Using an Ordinal Scale|Time to an Improvement of at Least Two Categories Using an Ordinal Scale|Time to Discharge or to a NEWS of 2 or Less and Maintained for 24 Hours, Whichever Occurs First
|
|
NCT04640168
|
Adaptive COVID-19 Treatment Trial 4 (ACTT-4) |
Completed |
Phase 3 |
Nov/24/2020 |
Jun/18/2021 |
- Alternative id - 20-0006 ACTT-4
- Interventions - Drug: Baricitinib|Drug: Dexamethasone|Other: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - University of Alabama at Birmingham School of Medicine - Infectious Disease, Birmingham, Alabama, United States|UCSF Fresno Center for Medical Education and Research - Clinical Research Center, Fresno, California, United States|University of California San Diego Health - Jacobs Medical Center, La Jolla, California, United States|University of California Los Angeles Medical Center - Westwood Clinic, Los Angeles, California, United States|University of California Irvine Medical Center - Infectious Disease, Orange, California, United States|VA Palo Alto Health Care System - Infectious Diseases, Palo Alto, California, United States|University of California Davis Medical Center - Internal Medicine - Infectious Disease, Sacramento, California, United States|Kaiser Permanente San Diego Medical Center, San Diego, California, United States|Naval Medical Center San Diego - Infectious Disease Clinic, San Diego, California, United States|University of California San Francisco - Zuckerberg San Francisco General Hospital - Division of Human Immunodeficiency Virus, Infectious Disease, and Global Medicine, San Francisco, California, United States|Stanford University - Stanford Hospital and Clinics - Pediatrics - Infectious Diseases, Stanford, California, United States|Cedars Sinai Medical Center, West Hollywood, California, United States|VA Eastern Colorado Health Care System, Aurora, Colorado, United States|Denver Health Division of Hospital Medicine - Main Campus, Denver, Colorado, United States|Georgetown University Medical Center - Division of Infectious Diseases, Washington, District of Columbia, United States|University of Florida Health - Shands Hospital - Division of Infectious Diseases and Global Medicine, Gainesville, Florida, United States|University of Florida Health - Jacksonville - Department of Emergency Medicine, Jacksonville, Florida, United States|University of Miami Miller School of Medicine - Infectious Diseases, Miami, Florida, United States|Emory Vaccine Center - The Hope Clinic, Decatur, Georgia, United States|Atlanta VA Medical Center - Infectious Diseases Clinic, Decatur, Georgia, United States|Tripler Army Medical Center, Honolulu, Hawaii, United States|Northwestern Hospital - Infectious Disease, Chicago, Illinois, United States|University of Illinois at Chicago College of Medicine - Division of Infectious Diseases, Chicago, Illinois, United States|University of Iowa Hospitals & Clinics - Department of Internal Medicine, Iowa City, Iowa, United States|Tulane University - Section of Pulmonary Diseases, Critical Care, and Environmental Medicine, New Orleans, Louisiana, United States|University of Maryland School of Medicine - Center for Vaccine Development - Baltimore, Baltimore, Maryland, United States|Johns Hopkins Hospital - Medicine - Infectious Diseases, Baltimore, Maryland, United States|Walter Reed National Military Medical Center, Bethesda, Maryland, United States|National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Clinical Research Section, Bethesda, Maryland, United States|Massachusetts General Hospital - Infectious Diseases, Boston, Massachusetts, United States|University of Massachusetts Medical School - Infectious Diseases and Immunology, Worcester, Massachusetts, United States|University of Michigan - Infectious Disease Clinic at Taubman Center, Ann Arbor, Michigan, United States|University of Minnesota Medical Center, Fairview - Infectious Diseases and International Medicine, Minneapolis, Minnesota, United States|Saint Louis University - Center for Vaccine Development, Saint Louis, Missouri, United States|University of Nebraska Medical Center - Infectious Diseases, Omaha, Nebraska, United States|CHI Health Creighton University Medical Center - Bergan Mercy - Pulmonary Medicine, Omaha, Nebraska, United States|Atlantic Health System - Morristown Medical Center, Morristown, New Jersey, United States|University of New Mexico Clinical and Translational Science Center, Albuquerque, New Mexico, United States|Montefiore Medical Center - Infectious Diseases, Bronx, New York, United States|New York University School of Medicine - Langone Medical Center - Microbiology - Parasitology, New York, New York, United States|University of Rochester Medical Center - Vaccine Research Unit, Rochester, New York, United States|Duke Human Vaccine Institute - Duke Vaccine and Trials Unit, Durham, North Carolina, United States|Womack Army Medical Center - Pulmonary and Respiratory Services, Fort Bragg, North Carolina, United States|University of Oklahoma Health Science Center - Surgery, Oklahoma City, Oklahoma, United States|Kaiser Permanente Northwest - Center for Health Research, Portland, Oregon, United States|Penn State Health Milton S. Hershey Medical Center - Division of Infectious Diseases, Hershey, Pennsylvania, United States|Hospital of the University of Pennsylvania - Infectious Diseases, Philadelphia, Pennsylvania, United States|University of Pittsburgh - Medicine - Infectious Diseases, Pittsburgh, Pennsylvania, United States|Baylor Scott & White Health - Baylor University Medical Center - North Texas Infectious Disease Consultants, Dallas, Texas, United States|University of Texas Southwestern Medical Center - Internal Medicine - Infectious Diseases, Dallas, Texas, United States|Brooke Army Medical Center, Fort Sam Houston, Texas, United States|University of Texas Medical Branch - Division of Infectious Disease, Galveston, Texas, United States|Methodist Hospital - Houston, Houston, Texas, United States|Baylor College of Medicine - Molecular Virology and Microbiology, Houston, Texas, United States|University of Texas Health Science Center at San Antonio - Infectious Diseases, San Antonio, Texas, United States|University of Utah - Infectious Diseases, Salt Lake City, Utah, United States|University of Virginia - Acute Care Surgery, Charlottesville, Virginia, United States|Naval Medical Center Portsmouth - Infectious Disease Division, Portsmouth, Virginia, United States|EvergreenHealth Infectious Disease Service, Kirkland, Washington, United States|Providence Sacred Heart Medical Center, Spokane, Washington, United States|Madigan Army Medical Center - Infectious Disease Clinic, Tacoma, Washington, United States|Tokyo Medical and Dental University - Medical Hospital - Department of Respiratory Medicine, Tokyo, Japan|National Center for Global Health and Medicine Hospital - Disease Control and Prevention Center, Tokyo, Japan|Seoul National University Bundang Hospital - Division of Infectious Diseases, Bundang-gu Seongnam-si, Gyeonggi-do, Korea, Republic of|Seoul National University Hospital, Seoul, Jongno-gu, Korea, Republic of|Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán - Departamento de Infectologia, Mexico City, Mexico|Instituto Nacional de Enfermedades Respiratorias (INER) - Ismael Cosío Villegas, Mexico City, Mexico|National University Health System - Division of Infectious Diseases, Singapore, Singapore|National University Health System - Alexandra Hospital - Division of Infectious Diseases, Singapore, Singapore|National Centre for Infectious Diseases, Singapore, Singapore|Changi General Hospital - Clinical Trials and Research Unit (CTRU), Singapore, Singapore|Ng Teng Fong General Hospital - Infectious Disease Service, Singapore, Singapore
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 1010
- Age - 18 Years to 99 Years (Adult, Older Adult)
- Outcome measures - The proportion of subjects not meeting criteria for one of the following two ordinal scale categories at any time: 8) Death; 7) Hospitalized, on invasive mechanical ventilation or extracorporeal membrane oxygenation (ECMO)|Change from baseline in alanine aminotransferase (ALT)|Change from baseline in aspartate aminotransferase (AST)|Change from baseline in C-reactive protein (CRP)|Change from baseline in creatinine|Change from baseline in d-dimer concentration|Change from baseline in glucose|Change from baseline in hemoglobin|Change from baseline in platelets|Change from baseline in prothrombin time (PT)|Change from baseline in total bilirubin|Change from baseline in white blood cell count (WBC) with differential|Cumulative incidence of Grade 3 and 4 clinical and/or laboratory adverse events (AEs)|Cumulative incidence of serious adverse events (SAEs)|Days of invasive mechanical ventilation/ extracorporeal membrane oxygenation (ECMO) (if applicable)|Days of non-invasive ventilation/high flow oxygen (if applicable)|Days of supplemental oxygen (if applicable)|Desirability of Outcome Ranking (DOOR)|Duration of hospitalization|Incidence of discontinuation or temporary suspension of study product administration|Subject 14-day mortality|Subject 28-day mortality|Subject clinical status|The proportion of subjects meeting criteria for each of the 8 ordinal scale categories|The proportion of subjects not meeting criteria for one of the three most severe ordinal scale categories at any time.|Time to an improvement of one category from baseline using an ordinal scale|Time to an improvement of two categories from baseline using an ordinal scale|Time to recovery
|
|
NCT04431453
|
Study to Evaluate the Safety, Tolerability, Pharmacokinetics, and Efficacy of Remdesivir (GS-5734™) in Participants From Birth to < 18 Years of Age With Coronavirus Disease 2019 (COVID-19) |
Recruiting |
Phase 2|Phase 3 |
Jul/21/2020 |
Feb/01/2022 |
- Alternative id - GS-US-540-5823|2020-001803-17
- Interventions - Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Children's Hospital of Alabama, Birmingham, Alabama, United States|Children's Hospital Los Angeles, Los Angeles, California, United States|Ronald Reagan University of California, Los Angeles Medical Center, Los Angeles, California, United States|Valley Children's Hospital, Madera, California, United States|Rady Children's Hospital San Diego, San Diego, California, United States|Tampa General Hospital (Inpatient Visits), Tampa, Florida, United States|Ann & Robert H. Lurie Children's Hospital, Chicago, Illinois, United States|Norton Children's Hospital, Louisville, Kentucky, United States|Tulane University School of Medicine, New Orleans, Louisiana, United States|Johns Hopkins Children's Center, Baltimore, Maryland, United States|Boston Children's Hospital, Boston, Massachusetts, United States|UMass Memorial Medical Center - University Campus, Worcester, Massachusetts, United States|Children's Hospital of Michigan, Detroit, Michigan, United States|Spectrum Health/Helen De Vos Children's Hospital, Grand Rapids, Michigan, United States|Children's Minnesota, Minneapolis, Minnesota, United States|Children's Hospital & Medical Center, Omaha, Nebraska, United States|NYC Health + Hospitals/Jacobi Medical Center, Bronx, New York, United States|Montefiore Medical Center, Bronx, New York, United States|Northwell Health-Cohen Children's Medical Center, New Hyde Park, New York, United States|Carolinas Medical Center-Levine Children's Hospital, Charlotte, North Carolina, United States|Lehigh Valley Hospital/Lehigh Valley Health Network (LVH/LVHN), Allentown, Pennsylvania, United States|St. Christopher's Hospital for Children, Philadelphia, Pennsylvania, United States|Children's Medical Center, Dallas, Texas, United States|Texas Children's Hospital, Houston, Texas, United States|Azienda Ospedaliero Universitaria Meyer, Florence, Italy|University Hospital of Padova, Clinic of Pediatric Hematology-Oncology, Padova, Italy|University Hospital of Parma, Parma, Italy|Hospital Universitari Vall D'Hebron, Barcelona, Spain|Hospital Sant Joan de Déu, Esplugues de llobregat, Spain|Hospital General Universitario Gregorio Maranon, Madrid, Spain|Hospital Universitario 12 de Octubre, Madrid, Spain|Hospital Universitario La Paz, Madrid, Spain|Hospital Clínico Universitario de Santiago, Santiago de Compostela, Spain|University Hospitals Birmingham NHS Foundation Trust, Birmingham, United Kingdom|Alder Hey Children's NHS Foundation Trust, Liverpool, United Kingdom|Guy's and St Thomas' NHS Foundation Trust, London, United Kingdom|King's College NHS Foundation Trust, London, United Kingdom
- Study designs - Allocation: N/A|Intervention Model: Single Group Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 52
- Age - up to 18 Years (Child, Adult)
- Outcome measures - Proportion of Participants Experiencing any Treatment-Emergent Adverse Events|Proportion of Participants Experiencing any Treatment-Emergent Graded Laboratory Abnormalities|Plasma Concentrations of Remdesivir (RDV) and Metabolites|Change From Baseline in Oxygenation Use|Change From Baseline in the Use of Mechanical Ventilation or Extracorporeal Membrane Oxygenation (ECMO)|Clinical Improvement on a 7-point Ordinal Scale|Time (days) to Discharge From Hospital|Days to First Confirmed Negative Polymerase Chain Reaction (PCR) Result|Change From Baseline in Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV-2) Viral Load|Bilirubin Concentrations in < 14-day-old Participants|Clinical Improvement Based on Scoring Using the Pediatric Early Warning Score (PEWS) Improvement Scale|Plasma Concentrations of Sulfobutylether β-cyclodextrin Sodium (SBECD)|Proportion of Participants With Concomitant Use of Medications other than RDV for Treatment of Coronavirus Disease 2019 (COVID-19)
|
|
NCT04501952
|
Study to Evaluate the Efficacy and Safety of Remdesivir (GS-5734™) Treatment of Coronavirus Disease 2019 (COVID-19) in an Outpatient Setting |
Terminated |
Phase 3 |
Sep/18/2020 |
May/06/2021 |
- Alternative id - GS-US-540-9012|2020-003510-12
- Interventions - Drug: RDV|Drug: Placebo to Match RDV
- Study type - Interventional
- Study results - Has Results
- Locations - Arizona Liver Health, Chandler, Arizona, United States|Arizona Clinical Trials, Tucson, Arizona, United States|St Joseph Hospital Eureka, Eureka, California, United States|St. Joseph Heritage Healthcare, Fullerton, California, United States|Elevated Health, Huntington Beach, California, United States|Ruane Clinical Research Group, Los Angeles, California, United States|LA Universal Center, INC., Los Angeles, California, United States|Mills Clinical Research, Los Angeles, California, United States|Kaiser Permanente Northern California, Oakland, California, United States|FOMAT Medical Research, Oxnard, California, United States|UC Davis Health, Sacramento, California, United States|Kaiser Permanente Northern California, 6600 Bruceville Road, Sacramento, California, United States|Kaiser Permanente Northern California, 2025 Morse Ave, Sacramento, California, United States|Kaiser Permanente Northern California, 1200 El Camino Real, San Francisco, California, United States|Kaiser Permanente Northern California, 2425 Geary Blvd, San Francisco, California, United States|UCSF Medical Center, San Francisco, California, United States|Kaiser Permanente Northern California, 250 Hospital Parkway, Suite 850, San Jose, California, United States|Kaiser Permanente Northern California, 2500 Merced St, San Leandro, California, United States|St. Joseph Heritage Healthcare, Santa Rosa, California, United States|Premiere Medical Center of Burbank, Inc, Toluca Lake, California, United States|Kaiser Permanente Northern California, 975 Sereno Drive, Vallejo, California, United States|New Hope Research Development DBA HCD, Whittier, California, United States|Centura Health Porter Place, Denver, Colorado, United States|Nuvance Health, Danbury, Connecticut, United States|RecioMed Clinical Research Network, Boynton Beach, Florida, United States|Midland Florida Clinical Research Center, LLC, DeLand, Florida, United States|Invesclinic, Fort Lauderdale, Florida, United States|Lawnwood Regional Medical Center, Fort Pierce, Florida, United States|Evolution Clinical Trials, Hialeah Gardens, Florida, United States|Encore Medical Research, Hollywood, Florida, United States|Advanced Pulmonary Research Institute, Loxahatchee Groves, Florida, United States|L&C Professional Medical Research Institute, Miami, Florida, United States|Laguna Clinical Research Associates, Miami, Florida, United States|CTMD Research, Inc, Palm Springs, Florida, United States|IMIC Inc, Palmetto Bay, Florida, United States|Luminous Clinical Research - South Florida Urgent Care, Pembroke Pines, Florida, United States|St. Josephs Comprehensive Research Institute, Tampa, Florida, United States|AIDS Research and Treatment Center of the Treasure Coast, Vero Beach, Florida, United States|Triple O Research Institute PA, West Palm Beach, Florida, United States|Agile Clinical Research Trials, Atlanta, Georgia, United States|Mercer University School of Medicine, Macon, Georgia, United States|Infectious Disease Associates of Kansas City, P.C.Infectious Disease Associates of Kansas City, P.C., Burr Ridge, Illinois, United States|Metro Infectious Disease Consultants, Burr Ridge, Illinois, United States|NorthStar Medical Center, Chicago, Illinois, United States|NorthShore University Healthsystem, Evanston, Illinois, United States|Tulane University, New Orleans, Louisiana, United States|Holy Cross Hospital, Inc., Baltimore, Maryland, United States|University of Maryland Baltimore, Baltimore, Maryland, United States|Tufts Medical Center, Boston, Massachusetts, United States|South Shore Hospital, South Weymouth, Massachusetts, United States|VA Boston Healthcare System, West Roxbury, Massachusetts, United States|Be Well Medical Center, Berkley, Michigan, United States|Onyx Research Institute, Flint, Michigan, United States|Memorial Hospital of Gulfport, Gulfport, Mississippi, United States|Metro Infectious Disease Consultants, Kansas City, Missouri, United States|Quality Clinical Research Inc., Omaha, Nebraska, United States|AB Clinical Trials, Las Vegas, Nevada, United States|AXCES Research Group, Santa Fe, New Mexico, United States|New York Presbyterian Hospital, Flushing, New York, United States|Northwell Health, New Hyde Park, New York, United States|Atrium Health Carolinas Medical Center, Charlotte, North Carolina, United States|East Carolina University, Greenville, North Carolina, United States|Rosedale Infectious Diseases, Huntersville, North Carolina, United States|Christ Hospital, Cincinnati, Ohio, United States|Cherokee Nation WW Hastings Hospital, Tahlequah, Oklahoma, United States|Providence St. Vincent Medical Center, Portland, Oregon, United States|Temple University, Philadelphia, Pennsylvania, United States|Avera Research Institute, Sioux Falls, South Dakota, United States|University of Tennessee Health Science Center, Knoxville, Tennessee, United States|Central Texas Clinical Research, Austin, Texas, United States|UT Physicians, Bellaire, Texas, United States|Baylor University Medical Center, 700 Scott and White Dr., College Station, Texas, United States|Baylor University Medical Center, Dallas, Texas, United States|UT Southwestern Medical Center, Dallas, Texas, United States|Care United Research, LLC, Forney, Texas, United States|VIP Trials, Harlingen, Texas, United States|University of Texas, Houston, Texas, United States|The Crofoot Research Center, Inc, Houston, Texas, United States|Baylor University Medical Center, 1901 North McArthur Blvd, Irving, Texas, United States|Laguna Clinical Research Associates, Laredo, Texas, United States|Diagnostic Clinic of Longview - Center for Clinical Research, Longview, Texas, United States|STAAMP Research, San Antonio, Texas, United States|Sugar Lakes Family Practice, Sugar Land, Texas, United States|Baylor University Medical Center, 2201 MacArthur Dr., Suite 100, Waco, Texas, United States|ClinPoint Trials, Waxahachie, Texas, United States|Intermountain Healthcare, Murray, Utah, United States|Virginia Commonwealth University, Richmond, Virginia, United States|Providence Regional Medical Center Everett, Everett, Washington, United States|Sound Medical Research, Port Orchard, Washington, United States|Fred Hutchinson Cancer Research Center, Seattle, Washington, United States|Providence Medical Research Center, Spokane, Washington, United States|Wisconsin Corporation for Biomedical Research, Milwaukee, Wisconsin, United States|Aalborg University Hospital, Aalborg, Denmark|Aarhus University Hospital, Aarhus N, Denmark|Rigshospitalet, Copenhagen, Denmark|Hvidovre Hospital, Hvidovre, Denmark|Odense University Hospital, Odense, Denmark|Hospital Universitari Germans Trias i Pujol, Badalona, Spain|Hospital Clinic, Barcelona, Spain|Hospital Universitario Infanta Leonor, Madrid, Spain|Bradford Teaching Hospitals NHS Foundation Trust, Bradford, United Kingdom|University College Hospital, London, United Kingdom|St Mary's Hospital, London, United Kingdom|Newcastle Upon Tyne Hospitals NHS Foundation Trust, Newcastle Upon Tyne, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 584
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures - Percentage of Participants With Coronavirus Disease 2019 (COVID-19) Related Hospitalization (Defined as at Least 24 Hours of Acute Care) or All-Cause Death by Day 28|Percentage of Participants Who Experienced Treatment-Emergent Adverse Events (TEAEs)|Percentage of Participants With COVID-19 Related Medical Visits Attended in Person by the Participant and a Health Care Professional (MAVs) or All-Cause Death by Day 28|Percentage of Participants Who Died by Day 28|Percentage of Participants With COVID-19 Related Hospitalization at Day 28|Percentage of Participants With COVID-19 Related Hospitalization or All-Cause Death by Day 14|Percentage of Participants With COVID-19 Related MAVs or All-Cause Death by Day 14|Time-Weighted Average Change in Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV-2) Viral Load From Baseline to Day 7|Time to Alleviation (Mild or Absent) of Baseline COVID-19 Symptoms as Reported on the COVID-19-adapted Influenza Patient-Reported Outcome Plus Questionnaire (FLU-PRO Plus)|Percentage of Participants With Worsening After Alleviation of Baseline COVID-19 Symptoms as Reported on the COVID-19-adapted FLU-PRO Plus Questionnaire|Percentage of Participants Who Required Oxygen Supplementation by Day 28
|
|
NCT04978259
|
SOLIDARITY Finland Long COVID-19 |
Recruiting |
Phase 4 |
Jul/24/2021 |
Dec/31/2023 |
- Alternative id - SOL21
- Interventions - Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - University of Helsinki, Helsinki, Finland
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 202
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Long-COVID symptoms|Quality of life (QoL)
|
|
NCT04401579
|
Adaptive COVID-19 Treatment Trial 2 (ACTT-2) |
Completed |
Phase 3 |
May/08/2020 |
Jul/31/2020 |
- Alternative id - 20-0006 ACTT-2
- Interventions - Other: Placebo|Drug: Remdesivir|Drug: Baricitinib
- Study type - Interventional
- Study results - Has Results
- Locations - University of Alabama at Birmingham School of Medicine - Infectious Disease, Birmingham, Alabama, United States|University of California San Diego Health - Jacobs Medical Center, La Jolla, California, United States|University of California Los Angeles Medical Center - Westwood Clinic, Los Angeles, California, United States|University of California Irvine Medical Center - Infectious Disease, Orange, California, United States|VA Palo Alto Health Care System - Infectious Diseases, Palo Alto, California, United States|Stanford University - Stanford Hospital and Clinics - Pediatrics - Infectious Diseases, Palo Alto, California, United States|University of California Davis Medical Center - Internal Medicine - Infectious Disease, Sacramento, California, United States|Naval Medical Center San Diego - Infectious Disease Clinic, San Diego, California, United States|University of California San Francisco - Zuckerberg San Francisco General Hospital - Division of Human Immunodeficiency Virus, Infectious Disease, and Global Medicine, San Francisco, California, United States|Cedars Sinai Medical Center, West Hollywood, California, United States|Eastern Colorado Health Care System, Aurora, Colorado, United States|Denver Health Division of Hospital Medicine - Main Campus, Denver, Colorado, United States|Georgetown University Medical Center - Division of Infectious Diseases, Washington, District of Columbia, United States|University of Florida Health - Shands Hospital - Division of Infectious Diseases and Global Medicine, Gainesville, Florida, United States|University of Miami Miller School of Medicine - Infectious Diseases, Miami, Florida, United States|Emory Vaccine Center - The Hope Clinic, Decatur, Georgia, United States|Atlanta VA Medical Center - Infectious Diseases Clinic, Decatur, Georgia, United States|Northwestern Hospital - Infectious Disease, Chicago, Illinois, United States|University of Illinois at Chicago College of Medicine - Division of Infectious Diseases, Chicago, Illinois, United States|Indiana University School of Medicine - Infectious Diseases, Indianapolis, Indiana, United States|Ochsner Medical Center - Kenner - Department of Infectious Diseases, Kenner, Louisiana, United States|Southeast Louisiana Veterans Health Care System (SLVHCS) - Section of Infectious Diseases, New Orleans, Louisiana, United States|University of Maryland School of Medicine - Center for Vaccine Development - Baltimore, Baltimore, Maryland, United States|Johns Hopkins Hospital - Medicine - Infectious Diseases, Baltimore, Maryland, United States|Walter Reed National Military Medical Center, Bethesda, Maryland, United States|National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Clinical Research Section, Bethesda, Maryland, United States|Massachusetts General Hospital - Infectious Diseases, Boston, Massachusetts, United States|University of Massachusetts Medical School - Infectious Diseases and Immunology, Worcester, Massachusetts, United States|University of Minnesota Medical Center, Fairview - Infectious Diseases and International Medicine, Minneapolis, Minnesota, United States|Saint Louis University - Center for Vaccine Development, Saint Louis, Missouri, United States|University of Nebraska Medical Center - Infectious Diseases, Omaha, Nebraska, United States|University of New Mexico Clinical and Translational Science Center, Albuquerque, New Mexico, United States|Montefiore Medical Center - Infectious Diseases, Bronx, New York, United States|New York University School of Medicine - Langone Medical Center - Microbiology - Parasitology, New York, New York, United States|University of Rochester Medical Center - Vaccine Research Unit, Rochester, New York, United States|Duke Human Vaccine Institute - Duke Vaccine and Trials Unit, Durham, North Carolina, United States|Womack Army Medical Center - Pulmonary and Respiratory Services, Fort Bragg, North Carolina, United States|Kaiser Permanente Northwest - Center for Health Research, Portland, Oregon, United States|Penn State Health Milton S. Hershey Medical Center - Division of Infectious Diseases, Hershey, Pennsylvania, United States|University of Pennsylvania Perelman School of Medicine - Penn Institute for Immunology, Philadelphia, Pennsylvania, United States|Vanderbilt University Medical Center - Infectious Diseases, Nashville, Tennessee, United States|Baylor Scott & White Health - Baylor University Medical Center - North Texas Infectious Disease Consultants, Dallas, Texas, United States|University of Texas Southwestern Medical Center - Internal Medicine - Infectious Diseases, Dallas, Texas, United States|Brooke Army Medical Center, Fort Sam Houston, Texas, United States|University of Texas Medical Branch - Division of Infectious Disease, Galveston, Texas, United States|Baylor College of Medicine - Molecular Virology and Microbiology, Houston, Texas, United States|University of Texas Health Science Center at San Antonio - Infectious Diseases, San Antonio, Texas, United States|University of Utah - Infectious Diseases, Salt Lake City, Utah, United States|University of Virginia - Acute Care Surgery, Charlottesville, Virginia, United States|Naval Medical Center Portsmouth - Infectious Disease Division, Portsmouth, Virginia, United States|EvergreenHealth Infectious Disease Service, Kirkland, Washington, United States|Providence Sacred Heart Medical Center, Spokane, Washington, United States|Madigan Army Medical Center - Infectious Disease Clinic, Tacoma, Washington, United States|University of Copenhagen - Centre of Excellence for Health, Immunity and Infections (CHIP) - Department of Infectious Diseases, Copenhagen, Denmark|National Center for Global Health and Medicine Hospital - Disease Control and Prevention Center, Tokyo, Japan|Seoul National University Bundang Hospital - Division of Infectious Diseases, Bundang-gu Seongnam-si, Korea, Republic of|Seoul National University Hospital, Seoul, Korea, Republic of|Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán - Departamento de Infectologia, Mexico City, Mexico|Instituto Nacional de Enfermedades Respiratorias (INER) - Ismael Cosío Villegas, Mexico City, Mexico|National University Health System - Division of Infectious Diseases, Singapore, Singapore|National Centre for Infectious Diseases (NCID), Singapore, Singapore|Changi General Hospital - Clinical Trials and Research Unit (CTRU), Singapore, Singapore|Ng Teng Fong General Hospital - Infectious Disease Service, Singapore, Singapore|Hospital Clinic Barcelona, Servicio de Salud Internacional, Barcelona, Cataluña, Spain|Hospital Germans Trias i Pujol - Servei Malalties Infeccioses, Barcelona, Cataluña, Spain|Hospital Clinico San Carlos - Enfermedades Infecciosas, Madrid, Spain|Royal Sussex County Hospital - Department of Intensive Care Medicine, Brighton, United Kingdom|Saint Thomas' Hospital - Directorate of Infection, City Of London, United Kingdom|St. James's University Hospital - Infectious Diseases, Leeds, United Kingdom|Royal Victoria Infirmary - Department of Infectious Diseases, Newcastle Upon Tyne, United Kingdom|John Radcliffe Hospital, Oxford, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 1033
- Age - 18 Years to 99 Years (Adult, Older Adult)
- Outcome measures - Time to Recovery|Time to Recovery by Race|Time to Recovery by Ethnicity|Time to Recovery by Sex|Change From Baseline in Alanine Transaminase (ALT)|Change From Baseline in Aspartate Transaminase (AST)|Change From Baseline in Creatinine|Change From Baseline in Glucose|Change From Baseline in Hemoglobin|Change From Baseline in Platelets|Change From Baseline in Prothrombin International Normalized Ratio (INR)|Change From Baseline in Total Bilirubin|Change From Baseline in White Blood Cell Count (WBC)|Change From Baseline in Neutrophils|Change From Baseline in Lymphocytes|Change From Baseline in Monocytes|Change From Baseline in Basophils|Change From Baseline in Eosinophils|Change in National Early Warning Score (NEWS) From Baseline|Percentage of Participants Reporting Grade 3 and 4 Clinical and/or Laboratory Adverse Events (AEs)|Percentage of Participants Reporting Serious Adverse Events (SAEs)|Duration of Hospitalization|Duration of New Non-invasive Ventilation or High Flow Oxygen Use|Duration of New Oxygen Use|Duration of New Ventilator or Extracorporeal Membrane Oxygenation (ECMO) Use|Duration of Oxygen Use|Percentage of Participants Discontinued or Temporarily Suspended From Investigational Therapeutics|Percentage of Participants Requiring New Ventilator or Extracorporeal Membrane Oxygenation (ECMO) Use|Percentage of Participants Requiring New Oxygen Use|Mean Change in the Ordinal Scale|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 15|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 1|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 3|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 5|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 8|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 11|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 22|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 29|14-day Participant Mortality|28-day Participant Mortality|Time to an Improvement of One Category Using an Ordinal Scale|Time to an Improvement of Two Categories Using an Ordinal Scale|Time to Discharge or to a NEWS of 2 or Less and Maintained for 24 Hours, Whichever Occurs First|Change From Baseline in C-reactive Protein (CRP)|Change From Baseline in D-dimer Concentration
|
|
NCT04492475
|
Adaptive COVID-19 Treatment Trial 3 (ACTT-3) |
Completed |
Phase 3 |
Aug/05/2020 |
Dec/21/2020 |
- Alternative id - 20-0006 ACTT-3
- Interventions - Drug: Interferon beta-1a|Other: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - Has Results
- Locations - University of Alabama at Birmingham School of Medicine - Infectious Disease, Birmingham, Alabama, United States|UCSF Fresno Center for Medical Education and Research - Clinical Research Center, Fresno, California, United States|University of California San Diego Health - Jacobs Medical Center, La Jolla, California, United States|University of California Los Angeles Medical Center - Westwood Clinic, Los Angeles, California, United States|University of California Irvine Medical Center - Infectious Disease, Orange, California, United States|VA Palo Alto Health Care System - Infectious Diseases, Palo Alto, California, United States|Stanford University - Stanford Hospital and Clinics - Pediatrics - Infectious Diseases, Palo Alto, California, United States|University of California Davis Medical Center - Internal Medicine - Infectious Disease, Sacramento, California, United States|Naval Medical Center San Diego - Infectious Disease Clinic, San Diego, California, United States|University of California San Francisco - Zuckerberg San Francisco General Hospital - Division of HIV, ID, and Global Medicine, San Francisco, California, United States|Cedars Sinai Medical Center, West Hollywood, California, United States|Eastern Colorado Health Care System, Aurora, Colorado, United States|Denver Health Division of Hospital Medicine - Main Campus, Denver, Colorado, United States|University of Florida Health - Shands Hospital - Division of Infectious Diseases and Global Medicine, Gainesville, Florida, United States|University of Florida Health - Jacksonville - Department of Emergency Medicine, Jacksonville, Florida, United States|University of Miami Miller School of Medicine - Infectious Diseases, Miami, Florida, United States|Emory Vaccine Center - The Hope Clinic, Decatur, Georgia, United States|Atlanta VA Medical Center - Infectious Diseases Clinic, Decatur, Georgia, United States|Tripler Army Medical Center (TAMC), Honolulu, Hawaii, United States|Northwestern Hospital - Infectious Disease, Chicago, Illinois, United States|University of Illinois at Chicago Division of Infectious Diseases, Chicago, Illinois, United States|University of Iowa Hospitals & Clinics - Department of Internal Medicine, Iowa City, Iowa, United States|Ochsner Medical Center - Kenner - Department of Infectious Diseases, Kenner, Louisiana, United States|Southeast Louisiana Veterans Health Care System - Section of Infectious Diseases, New Orleans, Louisiana, United States|University of Maryland School of Medicine - Center for Vaccine Development - Baltimore, Baltimore, Maryland, United States|Johns Hopkins Hospital - Medicine - Infectious Diseases, Baltimore, Maryland, United States|Walter Reed National Military Medical Center, Bethesda, Maryland, United States|National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Clinical Research Section, Bethesda, Maryland, United States|Massachusetts General Hospital - Infectious Diseases, Boston, Massachusetts, United States|University of Massachusetts Medical School - Infectious Diseases and Immunology, Worcester, Massachusetts, United States|University of Minnesota Medical Center, Fairview - Infectious Diseases and International Medicine, Minneapolis, Minnesota, United States|Saint Louis University - Center for Vaccine Development, Saint Louis, Missouri, United States|University of Nebraska Medical Center - Infectious Diseases, Omaha, Nebraska, United States|University of New Mexico Clinical and Translational Science Center, Albuquerque, New Mexico, United States|Montefiore Medical Center - Infectious Diseases, Bronx, New York, United States|New York University School of Medicine - Langone Medical Center - Microbiology - Parasitology, New York, New York, United States|University of Rochester Medical Center - Vaccine Research Unit, Rochester, New York, United States|Duke Human Vaccine Institute - Duke Vaccine and Trials Unit, Durham, North Carolina, United States|Womack Army Medical Center - Pulmonary and Respiratory Services, Fort Bragg, North Carolina, United States|Kaiser Permanente Northwest - Center for Health Research, Portland, Oregon, United States|Penn State Health Milton S. Hershey Medical Center - Division of Infectious Diseases, Hershey, Pennsylvania, United States|Hospital of the University of Pennsylvania - Infectious Diseases, Philadelphia, Pennsylvania, United States|Baylor Scott & White Health - Baylor University Medical Center - North Texas Infectious Disease Consultants, Dallas, Texas, United States|University of Texas Southwestern Medical Center - Internal Medicine - Infectious Diseases, Dallas, Texas, United States|Brooke Army Medical Center, Fort Sam Houston, Texas, United States|University of Texas Medical Branch - Division of Infectious Disease, Galveston, Texas, United States|Baylor College of Medicine - Molecular Virology and Microbiology, Houston, Texas, United States|Methodist Hospital - Houston, Houston, Texas, United States|University of Texas Health Science Center at San Antonio - Infectious Diseases, San Antonio, Texas, United States|University of Utah - Infectious Diseases, Salt Lake City, Utah, United States|University of Virginia - Acute Care Surgery, Charlottesville, Virginia, United States|Naval Medical Center Portsmouth - Infectious Disease Division, Portsmouth, Virginia, United States|EvergreenHealth Infectious Disease Service, Kirkland, Washington, United States|Providence Sacred Heart Medical Center, Spokane, Washington, United States|Madigan Army Medical Center - Infectious Disease Clinic, Tacoma, Washington, United States|National Center for Global Health and Medicine Hospital - Disease Control and Prevention Center, Tokyo, Japan|Seoul National University Bundang Hospital - Division of Infectious Diseases, Seongnam, Gyeonggi-do, Korea, Republic of|Seoul National University Hospital, Seoul, Jongno-gu, Korea, Republic of|Instituto Nacional de Enfermedades Respiratorias (INER) - Ismael Cosío Villegas, Mexico City, Mexico|Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán - Departamento de Infectologia, Mexico City, Mexico|National University Health System - Division of Infectious Diseases, Singapore, Singapore|National Centre for Infectious Diseases (NCID), Singapore, Singapore|Changi General Hospital - Clinical Trials and Research Unit (CTRU), Singapore, Singapore|Ng Teng Fong General Hospital - Infectious Disease Service, Singapore, Singapore
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 969
- Age - 18 Years to 99 Years (Adult, Older Adult)
- Outcome measures - Time to Recovery for Participants With Baseline Ordinal Score 4, 5 and 6|Time to Recovery for Participants With Baseline Ordinal Score 4, 5 and 6 by Race|Time to Recovery for Participants With Baseline Ordinal Score 4, 5 and 6 by Ethnicity|Time to Recovery for Participants With Baseline Ordinal Score 4, 5 and 6 by Sex|Change From Baseline in Alanine Aminotransferase (ALT)|Change From Baseline in Aspartate Aminotransferase (AST)|Change From Baseline in C-reactive Protein (CRP)|Change From Baseline in Creatinine|Change From Baseline in D-dimer Concentration|Change From Baseline in Hemoglobin|Change From Baseline in Prothrombin International Normalized Ratio (INR)|Change From Baseline in Platelets|Change From Baseline in Total Bilirubin|Change From Baseline in White Blood Cell Count (WBC)|Change From Baseline in Neutrophils|Change From Baseline in Lymphoctyes|Change From Baseline in Monocytes|Change From Baseline in Basophils|Change From Baseline in Eosinophils|Change in National Early Warning Score (NEWS) From Baseline|Percentage of Participants Reporting Grade 3 and 4 Clinical and/or Laboratory Adverse Events (AEs)|Percentage of Participants Reporting Serious Adverse Events (SAEs)|Duration of Hospitalization|Duration of Invasive Mechanical Ventilation|Duration of New Non-invasive Ventilation or High Flow Oxygen Use|Duration of New Oxygen Use|Duration of New Ventilator or Extracorporeal Membrane Oxygenation (ECMO) Use|Duration of Non Invasive Ventilation or High Flow Oxygen Use|Duration of Oxygen Use|Number of Participants With Discontinuation or Temporary Suspension of Study Product Administration|Proportion of Participants With New Non-invasive Ventilation or High Flow Oxygen Use|Proportion of Participants With New Oxygen Use|Proportion of Participants With New Ventilator or Extracorporeal Membrane Oxygenation (ECMO) Use|Mean Change From Baseline in the Ordinal Scale|Percentage of Participants at Each Clinical Status Using Ordinal Scale at Day 15 for Participants With Baseline Ordinal Score 4 and 5|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 1|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 3|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 5|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 8|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 11|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 15|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 22|Percentage of Participants Reporting Each Severity Rating on an 8 Point Ordinal Scale at Day 29|14-day Participant Mortality|28-day Participant Mortality|Time to an Improvement of One Category Using an Ordinal Scale|Time to an Improvement of Two Categories Using an Ordinal Scale|Time to Discharge or to a National Early Warning Score (NEWS) of
|
|
NCT04745351
|
Study to Evaluate the Efficacy and Safety of Remdesivir in Participants With Severely Reduced Kidney Function Who Are Hospitalized for Coronavirus Disease 2019 (COVID-19) |
Recruiting |
Phase 3 |
Mar/31/2021 |
Sep/01/2022 |
- Alternative id - GS-US-540-5912|2020-005416-22|DOH-27-012022-4779
- Interventions - Drug: Remdesivir|Drug: RDV Placebo|Drug: Standard of Care
- Study type - Interventional
- Study results - No Results Available
- Locations - St. Vincent's Health System, Birmingham, Alabama, United States|Children's Hospital of Alabama, Birmingham, Alabama, United States|UAB Hospital, Birmingham, Alabama, United States|Pulmonary Associates of Mobile, P.C., Mobile, Alabama, United States|St. Joseph Hospital Eureka, Eureka, California, United States|Hoag Memorial Hospital Presbyterian, 16200 Sand Canyon Ave, Irvine, California, United States|Hoag Memorial Hospital Presbyterian, Newport Beach, California, United States|Sutter Medical Center Sacramento, Sacramento, California, United States|San Francisco VA Medical Center, San Francisco, California, United States|Stanford Hospital and Clinics, Stanford, California, United States|Torrance Memorial Medical Center, Torrance, California, United States|MedStar Health Research Institute, Washington, District of Columbia, United States|George Washington Medical Faculty Associates, Washington, District of Columbia, United States|North Florida/ South Georgia Veterans Health System, Gainesville, Florida, United States|Mayo Clinic Florida, Jacksonville, Florida, United States|Genesis Clinical Research, Tampa, Florida, United States|Cleveland Clinic Florida, Weston, Florida, United States|Northwestern University Comprehensive Transplant Center, Chicago, Illinois, United States|University of Chicago, Chicago, Illinois, United States|Great Lakes Clinical Trials, Chicago, Illinois, United States|Northwestern Medicine Central DuPage Hospital, Winfield, Illinois, United States|Baptist Health Lexington, Lexington, Kentucky, United States|Rapides Regional Medical Center, Alexandria, Louisiana, United States|Tulane Medical Center, 2000 Canal St., New Orleans, Louisiana, United States|Tulane Medical Center, New Orleans, Louisiana, United States|Maine Medical Center, Portland, Maine, United States|Wake Forest University Health Sciences, Bethesda, Maryland, United States|Holy Cross Hospital, 19801 Observation Dr, Germantown, Maryland, United States|Holy Cross Hospital, Silver Spring, Maryland, United States|Massachusetts General Hospital, Boston, Massachusetts, United States|Newton-Wellesley Hospital, Newton, Massachusetts, United States|St. Clair Nephrology Research, Grosse Pointe Woods, Michigan, United States|G.V. (Sonny) Montgomery VAMC, Jackson, Michigan, United States|Mayo Clinic Hospital, Rochester, Minnesota, United States|University Medical Center of Southern Nevada, Las Vegas, Nevada, United States|Renown Regional Medical Center, Reno, Nevada, United States|Saint Michael's Medical Center, Newark, New Jersey, United States|University of New Mexico Hospital, Albuquerque, New Mexico, United States|Lincoln Medical Center, Bronx, New York, United States|Jacobi Medical Center, Bronx, New York, United States|Montefiore Medical Center, Bronx, New York, United States|New York - Presbyterian Hospital/Weill Cornell Medical Center, New York, New York, United States|Stony Brook University Hospital, Stony Brook, New York, United States|PMG Infectious Disease Consultants (administrative), Portland, Oregon, United States|Oregon Health and Science University, Portland, Oregon, United States|Temple University Hospital, Philadelphia, Pennsylvania, United States|Medical University of South Carolina, Charleston, South Carolina, United States|Baylor University Medical Center, Dallas, Texas, United States|UT Southwestern Medical Center - ID Clinical Research Unit, Dallas, Texas, United States|Houston Methodist Hospital, Houston, Texas, United States|Memorial Hermann Hospital at TMC, Houston, Texas, United States|University Hospital, San Antonio, Texas, United States|VCU Health Medical Center, Richmond, Virginia, United States|Providence Regional Medical Center Everett, Everett, Washington, United States|Swedish Medical Center, Seattle, Washington, United States|MultiCare Health System - Pulmonary and Critical Care Center, Spokane, Washington, United States|Providence Health Care, Spokane, Washington, United States|Providence Health Care, 5633 N Lidgerwood, Spokane, Washington, United States|MultiCare Good Samaritan Hospital, Tacoma, Washington, United States|Hospital Nossa Senhora das Gracas, Curitiba, Brazil|Hospital Alemao Oswaldo Cruz, Sao Paulo, Brazil|Fundacao Faculdade Regional de Medicina de Sao Jose do Rio Preto, São José do Rio Preto, Brazil|Hospital Garcia de Orta, Almada, Portugal|Centro Hospitalar Cova da Beira EPE, Covilhã, Portugal|Hospital Nélio Mendonça,, Funchal, Portugal|Hospital Senhora da Oliveira, Guimaraes, Portugal|Hospital da Luz, Lisboa, Portugal|Centro Hospitalar Lisboa Ocidental, Lisbon, Portugal|Centro Hospitalar do Porto - Hospital de Santo Antonio, Odivelas, Portugal|Centro Hospitalar de Vila Nova de Gaia, Vila Nova de Gaia, Portugal|George Regional Hospital, George, South Africa|Mediclinic Vergelegen, Somerset West, South Africa|Hospital Universitario Germans Trias I Pujol, Badalona, Spain|Hospital Universitario de Cruces, Barakaldo, Spain|Hospital del Mar, Barcelona, Spain|Hospital Universitari Vall D'Hebron, Barcelona, Spain|Hospital Universitari de Bellvitge, Barcelona, Spain|Hospital Clinic de Barcelona, Barcelona, Spain|Hospital General Universitario de Elche, Elche, Spain|Hospital General Universitario Gregorio Maranon, Madrid, Spain|Hospital Universitario 12 de Octubre, Madrid, Spain|Hospital General Universitario Reina Sofia, Murcia, Spain|Complejo Asistencial Universitario de Salamanca - H. Clinico, Salamanca, Spain|Hospital General Universitario de Valencia, Valencia, Spain|Hospital Universitari i Politecnic La Fe, Valencia, Spain|Barts Health NHS Trust, The Royal London Hospital, London, United Kingdom|Guy's & St Thomas Hospital, London, United Kingdom|Kings College Hospital, London, United Kingdom|Imperial College Healthcare NHS Trust, London, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 1116
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures - Composite of All-Cause Mortality or Invasive Mechanical Ventilation (IMV) Through Day 29|All-Cause Mortality Through Day 29|Invasive Mechanical Ventilation Through Day 29|Time to Recovery|Clinical Status Assessed by an 8-Point Ordinal Scale on Day 15|Clinical Status Assessed by an 8-Point Ordinal Scale on Day 29|Renal Replacement Therapy (RRT)-Free Days (Among Those Without End-Stage Kidney Disease [ESKD] at Randomization) Through Day 29|Recovery Through Day 29|Percentage of Participants Experiencing Serious Adverse Events (SAEs)|Percentage of Participants who Permanently Discontinue Investigational Drug due to Adverse Events (AEs)
|
|
NCT04292899
|
Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734™) in Participants With Severe Coronavirus Disease (COVID-19) |
Completed |
Phase 3 |
Mar/06/2020 |
Jun/30/2020 |
- Alternative id - GS-US-540-5773|2020-000841-15|ISRCTN15874265
- Interventions - Drug: Remdesivir|Drug: Standard of Care
- Study type - Interventional
- Study results - Has Results
- Locations - Kaiser Permanente Los Angeles Medical Center, 3340 E. La Palma Avenue, Anaheim, California, United States|Alta Bates Summit Medical Center, Berkeley, California, United States|Mills-Peninsula Medical Center, Burlingame, California, United States|Eden Medical Center, Castro Valley, California, United States|Kaiser Permanente Los Angeles Medical Center, 9333 Imperial Highway, Downey, California, United States|Kaiser Permanente Los Angeles Medical Center, 9961 Sierra Ave, Fontana, California, United States|St Joseph Hospital Eureka, Fortuna, California, United States|Kaiser Permanente Los Angeles Medical Center, 25825 S. Vermont Ave., Harbor City, California, United States|Kaiser Permanente Los Angeles Medical Center, Los Angeles, California, United States|Kaiser Permanente Los Angeles Medical Center, 6041 Cadillac Ave., Los Angeles, California, United States|Kaiser Permanente Los Angeles Medical Center, 5601 De Soto Avenue, Los Angeles, California, United States|Mission Hospital Regional Medical Center, Mission Viejo, California, United States|Kaiser Permanente Los Angeles Medical Center, 27300 Iris Avenue, Moreno Valley, California, United States|El Camino Hospital, Mountain View, California, United States|Hoag Memorial Hospital Presbyterian, Newport Beach, California, United States|Kaiser Permanente Oakland Medical Center, Oakland, California, United States|Kaiser Permanente Los Angeles Medical Center, 2295 S. Vineyard Avenue, Ontario, California, United States|The Center for Cancer Prevention and Treatment at St. Joseph Hospital of Orange, Orange, California, United States|Kaiser Permanente Los Angeles Medical Center, 13651 Willard Street, Panorama City, California, United States|Kaiser Permanente Los Angeles Medical Center, 10800 Magnolia Avenue, Riverside, California, United States|Sutter Medical Center Sacramento, One Medical Plaza, Roseville, California, United States|Sutter Medical Center Sacramento, Sacramento, California, United States|Kaiser Permanente Los Angeles Medical Center, 4647 Zion Avenue, San Diego, California, United States|Kaiser Permanente Los Angeles Medical Center, 9455 Clairemont Mesa Blvd, San Diego, California, United States|Kaiser Permanente Oakland Medical Center, 1200 El Camino Real, San Francisco, California, United States|California Pacific Medical Center-Infectious Disease Associates Medical Group, San Francisco, California, United States|Kaiser Permanente Oakland Medical Center, 2425 Geary Blvd, San Francisco, California, United States|Kaiser Permanente Oakland Medical Center, 250 Hospital Parkway, Suite 850, San Jose, California, United States|Kaiser Permanente Oakland Medical Center, 2500 Merced St, San Leandro, California, United States|Kaiser Permanente Oakland Medical Center, 700 Lawrence Expressway, Santa Clara, California, United States|Providence St. Johns Medical Center, Santa Monica, California, United States|Sutter Santa Rosa Regional Hospital, Santa Rosa, California, United States|Stanford Hospital, Stanford, California, United States|University of Colorado Denver, University of Colorado Hospital, Aurora, Colorado, United States|SCL Health St. Joseph Hospital, 1375 East 19th Ave, Denver, Colorado, United States|SCL Health St. Joseph Hospital, Denver, Colorado, United States|Rose Medical Center, Denver, Colorado, United States|SCL Health St. Joseph Hospital, 200 Exempla Circle., Lafayette, Colorado, United States|Yale-New Haven Hospital, New Haven, Connecticut, United States|Kaiser Permanente Hawaii Moanalua Medical Center, Honolulu, Hawaii, United States|John H. Stroger Jr. Hospital of Cook County, Chicago, Illinois, United States|Rush University Medical Center, Chicago, Illinois, United States|University of Chicago, Chicago, Illinois, United States|IU Health Methodist Hospital, Indianapolis, Indiana, United States|University of Iowa Hospitals & Clinics, Iowa City, Iowa, United States|Tulane University, New Orleans, Louisiana, United States|Maine Medical Center, Portland, Maine, United States|Holy Cross Hospital, Silver Spring, Maryland, United States|Tufts Medical Center, Boston, Massachusetts, United States|Brigham & Women's Hospital and Harvard Medical School, Boston, Massachusetts, United States|Beth Israel Deaconess Medical Center, Boston, Massachusetts, United States|The University of Michigan Hospitals and Health Systems, Ann Arbor, Michigan, United States|Henry Ford Health System, Detroit, Michigan, United States|Hennepin Healthcare, Minneapolis, Minnesota, United States|Mayo Clinic, Rochester, Minnesota, United States|Providence St Patrick Hospital and International Heart Institute of MT Foundation, Missoula, Montana, United States|Darmouth-Hitchhock Medical Center, Lebanon, New Hampshire, United States|Hackensack University Medical Center, Hackensack, New Jersey, United States|Robert Wood Johnson University Hospital Somerset, Hillsborough, New Jersey, United States|Robert Wood Johnson University Hospital Somerset, 1 RWJ Place, New Brunswick, New Jersey, United States|Robert Wood Johnson University Hospital Somerset, 201 Lyons Avenue, Newark, New Jersey, United States|St. Joseph's University Medical Center, Paterson, New Jersey, United States|Jacobi Medical Center, Bronx, New York, United States|James J. Peters Veterans Administration Medical Center, Bronx, New York, United States|Jamaica Hospital Medical Center, Jamaica, New York, United States|Danbury Hospital, Lagrangeville, New York, United States|North Shore University Hospital, Manhasset, New York, United States|North Shore University Hospital, 270-05 76th Ave, New Hyde Park, New York, United States|Icahn School of Medicine at Mount Sinai, 350 East 17th Street, New York, New York, United States|Icahn School of Medicine at Mount Sinai, 1000 10th Avenue, New York, New York, United States|Icahn School of Medicine at Mount Sinai, 440 West 114th St., New York, New York, United States|Icahn School of Medicine at Mount Sinai, New York, New York, United States|Columbia University Medical Center-New York Presbyterian Hospital, New York, New York, United States|New York Presbyterian Hospital/Weill Cornell Medical Center, New York, New York, United States|Duke University Medical Center, Durham, North Carolina, United States|University Hospitals Cleveland Medical Center, Cleveland, Ohio, United States|Kaiser Sunnyside Medical Center, 2875 NW Stucki Ave, Hillsboro, Oregon, United States|Providence Portland Medical Center, Portland, Oregon, United States|Providence St. Vincent Medical Center, Portland, Oregon, United States|Kaiser Sunnyside Medical Center, Portland, Oregon, United States|Hospital of the University of Pennsylvania, 51 N. 31st Street, Philadelphia, Pennsylvania, United States|Hospital of the University of Pennsylvania, Philadelphia, Pennsylvania, United States|Temple University Hospital, Philadelphia, Pennsylvania, United States|The Miriam Hospital, Providence, Rhode Island, United States|Prisma Health Richland Hospital, Columbia, South Carolina, United States|Prisma Health Richland Hospital, 701 Grove Road, Greenville, South Carolina, United States|The Liver Institute of Methodist Dallas Medical Center, Dallas, Texas, United States|UT Southwestern Medical Center Amelia Court, HIV Research Clinic, Dallas, Texas, United States|Baylor University Medical Center, Dallas, Texas, United States|UT Southwestern Medical Center Amelia Court, HIV Research Clinic, 5201 Harry Hines Blvd., Dallas, Texas, United States|UT Southwestern Medical Center Amelia Court, HIV Research Clinic, 6201 Harry Hines Blvd, Dallas, Texas, United States|Baylor University Medical Center, 1400 8th Ave, Fort Worth, Texas, United States|Houston Methodist Hospital, Houston, Texas, United States|Baylor University Medical Center, 2401 S. 31st St., Temple, Texas, United States|University of Utah Health, Salt Lake City, Utah, United States|Virginia Hospital Center, Arlington, Virginia, United States|Inova Fairfax Medical Campus, Falls Church, Virginia, United States|VCU Health Medical Center, Richmond, Virginia, United States|Providence Regional Medical Center Everett, Everett, Washington, United States|Kadlec Regional Medical Center, Kennewick, Washington, United States|Providence St. Peter Hospital, Olympia, Washington, United States|Virginia Mason Medical Center, Seattle, Washington, United States|Swedish Center for Comprehensive Care, Seattle, Washington, United States|MultiCare Deaconess Hospital, Spokane, Washington, United States|MultiCare Tacoma General Hospital, Tacoma, Washington, United States|St Joseph Medical Center, Tacoma, Washington, United States|The First Affiliated Hospital of Guangzhou Medical University, Guangzhou, China|CHU Pellegrin, Bordeaux, France|CHU de Montpellier-Hopital Gui de Chauliac, Montpelier Cedex 5, France|CHU de Nantes-Hotel Dieu, Nantes, France|Hopital Saint-Louis, Paris, France|Hopital Saint Antoine, Paris, France|Charite Universitatsmedizin Berlin, Campus Virchow-Klinikum, Medizinische Klinik Infektiologie Pneumologie, Berlin, Germany|Universitatsklinikum Dusseldorf, Klinik fur Gastroenterologie, Hepatologie und Infektiologie, Düsseldorf, Germany|Universitatsklinikum Hamburg-Eppendorf, Hamburg, Germany|Universitätsklinikum Schleswig-Holstein Campus Kiel, Kiel, Germany|Klinikum St. Georg gGmbH Klinik fur Infektiologie, Tropenmedizin, Nephrologie und Rheumatologie, Leipzig, Germany|Klinikum rechts der Isar der TU Munchen, Klinik und Poliklinik Innere Medizin 2, Munich, Germany|Klinik für Hämatologie, Onkologie, Immunologie, München, Germany|Robert-Bosch-Krankenhaus (RBK), Klinik Schillerhohe (KSH) und Dr. Margarete Fischer-Bosch-Institut fur klinische Pharmakologie (IKP), Stuttgart, Germany|Prince of Wales Hospital, Hong Kong, Hong Kong|Princess Margaret Hospital, Hong Kong, Hong Kong|Queen Mary Hospital, Hong Kong, Hong Kong|UOC Malattie Infettive, ASST Papa Giovanni XXIII, Bergamo, Italy|ASST degli Spedali Civili di Brescia, Brescia, Italy|ASST di Cremona - Azienda Socio Sanitaria Territor, Cremona, Italy|UOC Malattie Infettive, IRCCS Ospedale San Raffaele, Milano, Italy|Dipartimento di Malattie Infettive, Malattie Infettive I- Malattie Infettive III, AssT Fatebenefratelli Sacco Ospedale Luigi Sacco, Milano, Italy|UO Malattie Infettive, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, Milan, Italy|UOC Medicina Di Laboratorio, Azienda Ospedaliera di Padova, Padova PD, Italy|Azienda Ospedaliero Universitaria di Parma, Parma, Italy|UOC Malattie Infettive I, Fondazione IRCCS Policlinio San Matteo, Pavia, Italy|Ospedale Guglielmo da Saliceto AUSL di Piacenza, Piacenza, Italy|UOC Malattie Infettive ad Alta Intensita di Cure, Istituto Nazionale Malattie Infettive Lazzaro Spallanzani I.R.C.C.S., Roma, Italy|Clinica Universitaria Malattie Infettive, Ospedale Amedeo di Savoia, Torino, Italy|Yokohama Municipal Citizen's Hospital, Kanagawa, Japan|Nagoya City East Medical Center, Nagoya, Japan|Tokyo Metropolitan Bokutoh Hospital, Tokyo, Japan|Kyungpook National University Hospital, Daegu, Korea, Republic of|Seoul Medical Center, Seoul, Korea, Republic of|National Medical Center, Seoul, Korea, Republic of|Amsterdam University Medical Centre - Location AMC, Amsterdam, Netherlands|Leiden University Medical Center, Leiden, Netherlands|Erasmus Medical Centre, Rotterdam, Netherlands|National University Hospital, Singapore, Singapore|Singapore General Hospital, Singapore, Singapore|National Centre for Infectious Diseases, Tan Tock Seng Hospital, Singapore, Singapore|Hospital Principe de Asturias, Alcalá De Henares, Madrid, Spain|Complejo Hospitalario Universitario A Coruña, A Coruña, Spain|Hospital Universitari Vall d'Hebron, Barcelona, Spain|Hospital Universitari de Bellvitge, Barcelona, Spain|Hospital Clinic de Barcelona, Barcelona, Spain|Hospital Universitario Cruces, Bizkaia, Spain|Hospital Universitario Ramón y Cajal, Madrid, Spain|Hospital Universitario Fundación Jiménez Díaz, Madrid, Spain|Hospital Universitario 12 de Octubre, Madrid, Spain|Hospital Universitario La Paz, Madrid, Spain|Hospital Regional Universitario de Málaga, Málaga, Spain|Hospital Universitario Virgen del Rocio, Sevilla, Spain|Sahlgrenska University Hospital, Ostra, Gothenburg, Sweden|SUS (Skanes University Hospital), Malmo, Sweden|Karolinska University Hospital, Stockholm, Sweden|Hopitaux Universitaires de Genève, Genève 14, Switzerland|Ospedale Regionale di Locarno La Carità, Lugano, Switzerland|Universitätsspital Zürich, Zürich, Switzerland|Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan|China Medical University Hospital, Taichung, Taiwan|National Taiwan University Hospital, Taipei City, Taiwan|Imperial College Healthcare NHS Trust, London, Greater London, United Kingdom|Royal Infirmary of Edinburgh, Edinburgh, United Kingdom|Queen Elizabeth University Hospital, Glasgow, United Kingdom|Hull University Teaching Hospitals NHS Trust, Hull, United Kingdom|Royal Lancaster Hospital, Lancaster, United Kingdom|Liverpool University Hospital, Liverpool, United Kingdom|Northwick Park Hospital, London, United Kingdom|Royal Free London NHS Foundation Trust, London, United Kingdom|King's College Hospital NHS Trust, London, United Kingdom|University College London, London, United Kingdom|Manchester University NHS Foundation Trust, Manchester, United Kingdom|Manchester University NHS Foundation Trust, Manchester, United Kingdom|Pennine Acute Hospitals NHS Trust, Manchester, United Kingdom|Derriford Hospital, Plymouth, United Kingdom|Sheffield Teaching Hospitals, Sheffield, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 4891
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures - Part A: Percentage of Participants in Each Clinical Status Category as Assessed by a 7-Point Ordinal Scale on Day 14|Part A: Percentage of Participants Who Experienced Treatment-Emergent Adverse Events (TEAEs)
|
|
NCT04871633
|
Effectiveness of Remedesvir in COVID-19 Patients Presenting at Mayo Hospital Lahore |
Completed |
Not Applicable |
Aug/01/2020 |
Dec/30/2020 |
- Alternative id - 483/RC/KEMU
- Interventions - Drug: Remdesivir|Drug: Conventional
- Study type - Interventional
- Study results - No Results Available
- Locations - Mayo Hospital, Lahore, Punjab, Pakistan
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 66
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures - Hospital Stay|Ventilation
|
|
NCT04292730
|
Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734™) in Participants With Moderate Coronavirus Disease (COVID-19) Compared to Standard of Care Treatment |
Completed |
Phase 3 |
Mar/15/2020 |
Jun/26/2020 |
- Alternative id - GS-US-540-5774|2020-000842-32|ISRCTN85762140
- Interventions - Drug: Remdesivir|Drug: Standard of Care
- Study type - Interventional
- Study results - Has Results
- Locations - Kaiser Permanente Los Angeles Medical Center, 3340 E. La Palma Avenue, Anaheim, California, United States|Alta Bates Summit Medical Center, Berkeley, California, United States|Mills-Peninsula Medical Center, Burlingame, California, United States|Eden Medical Center, Castro Valley, California, United States|Kaiser Permanente Los Angeles Medical Center, 9333 Imperial Highway, Downey, California, United States|Kaiser Permanente Los Angeles Medical Center, 9961 Sierra Ave, Fontana, California, United States|St Joseph Hospital Eureka, Fortuna, California, United States|Kaiser Permanente Los Angeles Medical Center, 25825 S. Vermont Ave., Harbor City, California, United States|Kaiser Permanente Los Angeles Medical Center, Los Angeles, California, United States|Kaiser Permanente Los Angeles Medical Center, 6041 Cadillac Ave., Los Angeles, California, United States|Kaiser Permanente Los Angeles Medical Center, 5601 De Soto Avenue, Los Angeles, California, United States|Mission Hospital Regional Medical Center, Mission Viejo, California, United States|Kaiser Permanente Los Angeles Medical Center,27300 Iris Avenue, Moreno Valley, California, United States|El Camino Hospital, Mountain View, California, United States|Hoag Memorial Hospital Presbyterian, Newport Beach, California, United States|Kaiser Permanente Oakland Medical Center, Oakland, California, United States|Kaiser Permanente Los Angeles Medical Center, 2295 S. Vineyard Avenue, Ontario, California, United States|The Center for Cancer Prevention and Treatment at St. Joseph Hospital of Orange, Orange, California, United States|Kaiser Permanente Los Angeles Medical Center,13651 Willard Street, Panorama City, California, United States|Kaiser Permanente Los Angeles Medical Center, 10800 Magnolia Avenue, Riverside, California, United States|Sutter Medical Center Sacramento, One Medical Plaza, Roseville, California, United States|Sutter Medical Center Sacramento, Sacramento, California, United States|Kaiser Permanente Los Angeles Medical Center, 4647 Zion Avenue, San Diego, California, United States|Kaiser Permanente Los Angeles Medical Center, 9455 Clairemont Mesa Blvd, San Diego, California, United States|Kaiser Permanente Oakland Medical Center, 1200 El Camino Real, San Francisco, California, United States|California Pacific Medical Center-Infectious Disease Associates Medical Group, San Francisco, California, United States|Kaiser Permanente Oakland Medical Center, 2425 Geary Blvd, San Francisco, California, United States|Kaiser Permanente Oakland Medical Center, 250 Hospital Parkway, Suite 850, San Jose, California, United States|Kaiser Permanente Oakland Medical Center, 2500 Merced St, San Leandro, California, United States|Kaiser Permanente Oakland Medical Center, 700 Lawrence Expressway, Santa Clara, California, United States|Providence St. Johns Medical Center, Santa Monica, California, United States|Sutter Santa Rosa Regional Hospital, Santa Rosa, California, United States|Stanford Hospital, Stanford, California, United States|University of Colorado Denver, University of Colorado Hospital, Aurora, Colorado, United States|SCL Health St. Joseph Hospital, 1375 East 19th Ave, Denver, Colorado, United States|SCL Health St. Joseph Hospital, Denver, Colorado, United States|Rose Medical Center, Denver, Colorado, United States|SCL Health St. Joseph Hospital, 200 Exempla Circle., Lafayette, Colorado, United States|Yale University, New Haven, Connecticut, United States|Kaiser Permanente Hawaii Moanalua Medical Center, Honolulu, Hawaii, United States|Cook County General Hospital, Chicago, Illinois, United States|Rush University Medical Center, Chicago, Illinois, United States|University of Chicago, Chicago, Illinois, United States|IU Health Methodist Hospital, Indianapolis, Indiana, United States|University of Iowa Hospitals & Clinics, Iowa City, Iowa, United States|Tulane University, New Orleans, Louisiana, United States|Maine Medical Center, Portland, Maine, United States|Holy Cross Hospital, Silver Spring, Maryland, United States|Tufts Medical Center, Boston, Massachusetts, United States|Brigham & Women's Hospital and Harvard Medical School, Boston, Massachusetts, United States|Center for Virology and Vaccine Research, Beth Israel Deaconess Medical Center, Boston, Massachusetts, United States|The University of Michigan Hospitals and Health Systems, Ann Arbor, Michigan, United States|Henry Ford Health System, Detroit, Michigan, United States|Hennepin Healthcare, Minneapolis, Minnesota, United States|Mayo Clinic, Rochester, Minnesota, United States|Providence St Patrick Hospital and International Heart Institute of MT Foundation, Missoula, Montana, United States|Darmouth-Hitchhock Medical Center, Lebanon, New Hampshire, United States|Hackensack Medical Center, Hackensack, New Jersey, United States|Robert Wood Johnson University Hospital Somerset, Hillsborough, New Jersey, United States|Robert Wood Johnson University Hospital Somerset, 1 RWJ Place, New Brunswick, New Jersey, United States|Robert Wood Johnson University Hospital Somerset, 201 Lyons Avenue, Newark, New Jersey, United States|St. Joseph's University Medical Center, Paterson, New Jersey, United States|Jacobi Medical Center, Bronx, New York, United States|James J. Peters Veterans Administration Medical Center, Bronx, New York, United States|Jamaica Hospital Medical Center, Jamaica, New York, United States|Danbury Hospital, Lagrangeville, New York, United States|North Shore University Hospital, Manhasset, New York, United States|North Shore University Hospital, 270-05 76th Ave, New Hyde Park, New York, United States|Icahn School of Medicine at Mount Sinai, 350 East 17th Street, New York, New York, United States|Icahn School of Medicine at Mount Sinai, 1000 10th Avenue, New York, New York, United States|Icahn School of Medicine at Mount Sinai, 440 West 114th St., New York, New York, United States|Icahn School of Medicine at Mount Sinai, New York, New York, United States|Columbia University Irving Medical Center, New York, New York, United States|Weill Cornell Medical College/NYU Presbyterian Hospital, New York, New York, United States|Duke University Medical Center, Durham, North Carolina, United States|University Hospitals Cleveland Medical Center, Cleveland, Ohio, United States|Kaiser Sunnyside Medical Center, 2875 NW Stucki Ave, Hillsboro, Oregon, United States|Providence Portland Medical Center, Portland, Oregon, United States|Providence St. Vincent Medical Center, Portland, Oregon, United States|Kaiser Sunnyside Medical Center, Portland, Oregon, United States|Hospital of the University of Pennsylvania, 51 N. 31st Street, Philadelphia, Pennsylvania, United States|Hospital of the University of Pennsylvania, Philadelphia, Pennsylvania, United States|Temple University Hospital, Philadelphia, Pennsylvania, United States|The Miriam Hospital, Providence, Rhode Island, United States|Prisma Health Richland Hospital, Columbia, South Carolina, United States|Prisma Health Richland Hospital, 701 Grove Road, Greenville, South Carolina, United States|The Liver Institute of Methodist Dallas Medical Center, Dallas, Texas, United States|UT Southwestern Medical Center Amelia Court, HIV Research Clinic, Dallas, Texas, United States|Baylor University Medical Center, Dallas, Texas, United States|UT Southwestern Medical Center Amelia Court, HIV Research Clinic, 5201 Harry Hines Blvd., Dallas, Texas, United States|UT Southwestern Medical Center Amelia Court, HIV Research Clinic, 6201 Harry Hines Blvd, Dallas, Texas, United States|Baylor University Medical Center, 1400 8th Ave, Fort Worth, Texas, United States|Houston Methodist Hospital, Houston, Texas, United States|Baylor University Medical Center, 2401 S. 31st St., Temple, Texas, United States|University of Utah Health, Salt Lake City, Utah, United States|Virginia Hospital Center, Arlington, Virginia, United States|Inova Fairfax Hospital, Falls Church, Virginia, United States|VCU Health Medical Center, Richmond, Virginia, United States|Providence Medical Research Center, Everett, Washington, United States|Kadlec Regional Medical Center, Kennewick, Washington, United States|Providence St. Peter Hospital, Olympia, Washington, United States|Virginia Mason Medical Center, Seattle, Washington, United States|Swedish Center for Comprehensive Care, Seattle, Washington, United States|MultiCare Deaconess Hospital, Spokane, Washington, United States|MultiCare Tacoma General Hospital, Tacoma, Washington, United States|St Joseph Medical Center, Tacoma, Washington, United States|The First Affiliated Hospital of Guangzhou Medical University, Guangzhou, China|CHU Pellegrin, Bordeaux, France|CHU de Montpellier-Hopital Gui de Chauliac, Montpelier Cedex 5, France|CHU de Nantes-Hotel Dieu, Nantes, France|Hopital Saint-Louis, Paris, France|Hopital Saint Antoine, Paris, France|Charite Universitatsmedizin Berlin, Campus Virchow-Klinikum, Medizinische Klinik Infektiologie Pneumologie, Berlin, Germany|Universitatsklinikum Dusseldorf, Klinik fur Gastroenterologie, Hepatologie und Infektiologie, Düsseldorf, Germany|Universitatsklinikum Hamburg-Eppendorf, Hamburg, Germany|Universitätsklinikum Schleswig-Holstein Campus Kiel, Kiel, Germany|Klinikum St. Georg gGmbH Klinik fur Infektiologie, Tropenmedizin, Nephrologie und Rheumatologie, Leipzig, Germany|Klinikum rechts der Isar der TU Munchen, Klinik und Poliklinik Innere Medizin 2, Munich, Germany|Klinik für Hämatologie, Onkologie, Immunologie, München, Germany|Robert-Bosch-Krankenhaus (RBK), Klinik Schillerhohe (KSH) und Dr. Margarete Fischer-Bosch-Institut fur klinische Pharmakologie (IKP), Stuttgart, Germany|Prince of Wales Hospital, Hong Kong, Hong Kong|Princess Margaret Hospital, Hong Kong, Hong Kong|Queen Mary Hospital, Hong Kong, Hong Kong|UOC Malattie Infettive, ASST Papa Giovanni XXIII, Bergamo, Italy|ASST degli Spedali Civili di Brescia, Brescia, Italy|ASST di Cremona - Azienda Socio Sanitaria Territor, Cremona, Italy|UOC Malattie Infettive, IRCCS Ospedale San Raffaele, Milano, Italy|ASST Fatebenefratelli Sacco, Milano, Italy|UO Malattie Infettive, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, Milan, Italy|UOC Malattie Infettive e Tropicali, Azienda Ospedaliera di Padova, Padova PD, Italy|Azienda Ospedaliero Universitaria di Parma, Parma, Italy|Malattie Infettive I, Fondazione IRCCS Policlinico San Matteo, Pavia, Italy|Ospedale Guglielmo da Saliceto AUSL di Piacenza, Piacenza, Italy|UOC Malattie Infettive ad Alta Intensita di Cure, Istituto Nazionale Malattie Infettive Lazzaro Spallanzani I.R.C.C.S., Roma, Italy|Clinica Universitaria Malattie Infettive, Ospedale Amedeo di Savoia, Torino, Italy|Yokohama Municipal Citizen's Hospital, Kanagawa, Japan|Nagoya City East Medical Center, Nagoya, Japan|Tokyo Metropolitan Bokutoh Hospital, Tokyo, Japan|Kyungpook National University Hospital, Daegu, Korea, Republic of|Seoul Medical Center, Seoul, Korea, Republic of|National Medical Center, Seoul, Korea, Republic of|Amsterdam University Medical Centre - Location AMC, Amsterdam, Netherlands|Leiden University Medical Center, Leiden, Netherlands|Erasmus Medical Centre, Rotterdam, Netherlands|National University Hospital, Singapore, Singapore|Singapore General Hospital, Singapore, Singapore|National Centre for Infectious Diseases, Tan Tock Seng Hospital, Singapore, Singapore|Hospital Principe de Asturias, Alcalá De Henares, Madrid, Spain|Complejo Hospitalario Universitario A Coruña, A Coruña, Spain|Hospital Universitario Vall d'Hebron, Barcelona, Spain|Hospital Universitari de Bellvitge, Barcelona, Spain|Hospital Clinic de Barcelona, Barcelona, Spain|Hospital Universitario Cruces, Bizkaia, Spain|Hospital Universitario Ramón y Cajal, Madrid, Spain|Hospital Universitario Fundación Jiménez Díaz, Madrid, Spain|Hospital Universitario 12 de Octubre, Madrid, Spain|Hospital Universitario La Paz, Madrid, Spain|Hospital Regional Universitario de Málaga, Málaga, Spain|Hospital Universitario Virgen del Rocio, Sevilla, Spain|Hospital Universitari i Politecnic La Fe, Valencia, Spain|Sahlgrenska University Hospital, Ostra, Gothenburg, Sweden|SUS (Skanes University Hospital), Malmo, Sweden|Karolinska University Hospital, Stockholm, Sweden|Hopitaux Universitaires de Genève, Genève 14, Switzerland|Ospedale Regionale di Locarno La Carità, Lugano, Switzerland|Universitätsspital Zürich, Zürich, Switzerland|Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan|China Medical University Hospital, Taichung, Taiwan|National Taiwan University Hospital, Taipei City, Taiwan|Imperial College NHS Trust, London, Greater London, United Kingdom|NHS Lothian, Royal lnfirmary of Edinburgh, Edinburgh, United Kingdom|Queen Elizabeth University Hospital, Glasgow, United Kingdom|Hull University Teaching Hospitals NHS Trust, Hull, United Kingdom|Royal Lancaster Hospital, Lancaster, United Kingdom|Liverpool University Hospital, Liverpool, United Kingdom|Northwick Park Hospital, London, United Kingdom|Royal Free London NHS Foundation Trust, London, United Kingdom|King's College Hospital NHS Trust, London, United Kingdom|University College London, London, United Kingdom|Manchester University NHS Foundation Trust, Manchester, United Kingdom|Manchester University NHS Foundation Trust, Manchester, United Kingdom|Pennine Acute Hospitals NHS Trust, Manchester, United Kingdom|Derriford Hospital, Plymouth, United Kingdom|Sheffield Teaching Hospitals, Sheffield, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 1113
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures - Part A: Percentage of Participants in Each Clinical Status Category as Assessed by a 7-Point Ordinal Scale on Day 11|Part A: Percentage of Participants Who Experienced Treatment-Emergent Adverse Events (TEAEs)
|
|
NCT04501978
|
ACTIV-3: Therapeutics for Inpatients With COVID-19 |
Active, not recruiting |
Phase 3 |
Aug/04/2020 |
Jul/01/2022 |
- Alternative id - 014 / ACTIV-3
- Interventions - Biological: LY3819253|Drug: Placebo|Biological: Remdesivir|Biological: VIR-7831|Biological: BRII-196/BRII-198|Biological: AZD7442|Drug: MP0420|Drug: PF-07304814
- Study type - Interventional
- Study results - No Results Available
- Locations - Banner University Medical Center Tucson (Site 206-004), 1625 N. Campbell Avenue, Tucson, Arizona, United States|Southern Arizona VA Healthcare System (Site 074-009), 3601 S. 6th Ave., Tucson, Arizona, United States|Velocity Chula Vista (Site 080-034), 752 Medical Center Ct., Ste. 304, Chula Vista, California, United States|Community Regional Medical Center (Site 203-005), 2823 Fresno Street, Fresno, California, United States|Velocity San Diego (Site 080-035), 5565 Grossmont Center Drive, Building 2, Suite 1, La Mesa, California, United States|VA Loma Linda Healthcare System (Site 074-017), 11201 Benton Street, Loma Linda, California, United States|VA Long Beach Healthcare System (Site 074-026), 5901 East 7th Street (09/151-M2), Long Beach, California, United States|Keck Hospital of USC (Site 301-020), 1500 San Pablo Street, Los Angeles, California, United States|Cedars-Sinai Medical Center (Site 208-002), 8700 Beverly Blvd., Los Angeles, California, United States|Ronald Reagan UCLA Medical Center (Site 203-002), 757 Westwood Plaza, Los Angeles, California, United States|Sacramento VA Medical Center (Site 074-023), 10535 Hospital Way, Mather, California, United States|Hoag Memorial Hospital Presbyterian (Site 080-026), One Hoag Drive, Newport Beach, California, United States|Palo Alto VAMC (Site 074-005), 3801 Miranda Avenue, Palo Alto, California, United States|UC Davis Health (Site 203-004), 2315 Stockton Blvd., Sacramento, California, United States|VA San Diego Healthcare System (Site 074-016), 3350 La Jolla Village Drive, San Diego, California, United States|UCSF Medical Center at Mount Zion (Site 203-007), 1600 Divisadero St., San Francisco, California, United States|San Francisco VAMC (Site 074-002), 4150 Clement St., San Francisco, California, United States|UCSF Medical Center (Site 203-001), Moffitt-Long Hospital, 505 Parnassus Ave., San Francisco, California, United States|Stanford University Hospital & Clinics (Site 203-003), 300 Pasteur Dr., Stanford, California, United States|Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center (Site 066-002), 1124 W. Carson Street, CDCRC Building, Torrance, California, United States|University of Colorado Hospital (Site 204-001), 12605 E. 16th Avenue, Aurora, Colorado, United States|Denver Public Health (Site 017-004), 660 Bannock St., MC2600 (Infectious Disease Clinic), Denver, Colorado, United States|National Jewish Health / St. Joseph Hospital (Site 204-003), 1400 Jackson Street, Denver, Colorado, United States|West Haven VA Medical Center (Site 025-007), 950 Campbell Avenue, West Haven, Connecticut, United States|MedStar Georgetown University Hospital (Site 067-001), 3800 Reservoir Road NW, Washington, District of Columbia, United States|MedStar Health Research Institute (Site 009-021), MedStar Washington Hospital Center, 110 Irving St., NW., Washington, District of Columbia, United States|Washington DC VA Medical Center (Site 009-004), 50 Irving Street NW, Washington, District of Columbia, United States|Bay Pines VAMC (Site 074-004), 10000 Bay Pines Blvd., Bldg. 100, Room 5B-104, Bay Pines, Florida, United States|Baycare Health System (Site 301-025), Morton Plant Hospital, 300 Pinellas Street, Clearwater, Florida, United States|North Florida/South Georgia Veterans Health System (Site 074-011), 1601 SW. Archer Road, Gainesville, Florida, United States|Memorial Healthcare System (Site 648-002), Memorial Regional Hospital, 3501 Johnson Street, Hollywood, Florida, United States|Miami VAMC (Site 074-003), 1201 NW 16 Street, Miami, Florida, United States|Hillsborough County Health Department, University of South Florida (Site 032-001), Tampa, Florida, United States|Emory University (Site 301-008), The Emory Clinic, Bldg. A, Suite 2236, 1365 Clifton Rd., NE, Atlanta, Georgia, United States|Lutheran Medical Group (Site 301-010), 7916 W. Jefferson Boulevard, Fort Wayne, Indiana, United States|Cotton O'Neil Clinical Research Center (Site 080-030), Stormont Vail Health, 1500 SW 10th Avenue, Topeka, Kansas, United States|University of Kentucky Hospital (Site 210-004), 1000 South Limestone St., Lexington, Kentucky, United States|Ochsner Clinic Foundation (Site 301-015), 1514 Jefferson Highway, New Orleans, Louisiana, United States|University of Maryland Medical Center (Site 301-019), 22 South Greene Street, Baltimore, Maryland, United States|Massachusetts General Hospital (Site 202-002), 55 Fruit Street, Boston, Massachusetts, United States|Beth Israel Deaconess Medical Center (Site 202-001), 330 Brookline Ave., Boston, Massachusetts, United States|Baystate Medical Center (Site 201-001), 759 Chestnut Street, Springfield, Massachusetts, United States|University of Michigan (Site 205-001), 1500 East Medical Center Drive, Ann Arbor, Michigan, United States|Henry Ford Health System, Henry Ford Hospital (Site 014-001), 2799 W. Grand Blvd., Detroit, Michigan, United States|Minneapolis Heart Institute Foundation (Site 301-026), Abbott Northwestern Hospital, 920 E 28th St. #100, Minneapolis, Minnesota, United States|Hennepin Healthcare (Site 027-001), 701 Park Avenue, Minneapolis, Minnesota, United States|Minneapolis VA Health Care System (Site 105-001), 1 Veterans Drive, Bldg 70, Minneapolis, Minnesota, United States|M Health Fairview University of Minnesota Medical Center (Site 112-001), 500 Harvard St. SE., Minneapolis, Minnesota, United States|University of Mississippi Medical Center (Site 202-005), 2500 North State Street, Jackson, Mississippi, United States|VA St. Louis Healthcare System (Site 074-027), 915 North Grand Blvd., Rm. C201, Saint Louis, Missouri, United States|Dartmouth-Hitchcock Medical Center/Mary Hitchcock Memorial Hospital (Site 301-024), One Medical Center Drive, Lebanon, New Hampshire, United States|Cooper University Hospital (Site 019-001), One Cooper Plaza, Camden, New Jersey, United States|Lincoln Medical Center (New York Health and Hospitals/Lincoln) (Site 003-016), 234 E. 149th Street, Bronx, New York, United States|Montefiore Medical Center Weiler Hospital (Site 206-003), 1825 Eastchester Road, Bronx, New York, United States|Montefiore Medical Center Moses Hospital (Site 206-001), 111 E. 210th Street, Bronx, New York, United States|SUNY Downstate Medical Center (Site 033-001), 450 Clarkson Ave., Brooklyn, New York, United States|Maimonides Medical Center (Site 033-002), 4802 10th Avenue, Brooklyn, New York, United States|Ichan School of Medicine at Mount Sinai (Site 301-012), One Gustave L. Levy Place, Box 1620, New York, New York, United States|Duke University Hospital (Site 301-006), 2301 Erwin Road, Durham, North Carolina, United States|Wake Forest University Health Sciences (Site 210-001), Medical Center Blvd, Winston-Salem, North Carolina, United States|University of Cincinnati Medical Center (Site 207-003), 234 Goodman Ave., Cincinnati, Ohio, United States|University Hospitals Cleveland Medical Center (Site 108-001), 11100 Euclid Avenue, Cleveland, Ohio, United States|Cleveland Clinic Fairview Hospital (Site 207-005), 18101 Lorain Avenue, Cleveland, Ohio, United States|Cleveland Clinic Foundation (Site 207-001), 9500 Euclid Avenue, Cleveland, Ohio, United States|Cleveland Clinic Marymount Hospital (Site 207-006), 12300 McCraken Road, Garfield Heights, Ohio, United States|Oregon Health & Science University (Site 208-003), 3181 SW Sam Jackson Park Rd., Portland, Oregon, United States|Portland VA Healthcare System (Site 074-024), 3710 SW. US Veterans Hospital Road, Portland, Oregon, United States|UPMC Magee-Womens Hospital (Site 209-003), 300 Halket Street, Pittsburgh, Pennsylvania, United States|UPMC Presbyterian Hospital (Site 209-001), 200 Lothrop Street, Pittsburgh, Pennsylvania, United States|UPMC Shadyside Hospital (Site 209-005), 5230 Centre Avenue, Pittsburgh, Pennsylvania, United States|Rhode Island Hospital (Site 080-036), 593 Eddy Street, Providence, Rhode Island, United States|The Miriam Hospital (Site 080-039), 164 Summit Ave., Providence, Rhode Island, United States|VA Providence Healthcare System (Site 074-025), 830 Chalkstone Ave., Providence, Rhode Island, United States|Ralph H. Johnson VA Medical Center (Site 074-015), 109 Bee Street, Charleston, South Carolina, United States|MUSC Research Nexus Clinic (Site 210-002), 96 Jonathan Lucas St., CSB 214, Charleston, South Carolina, United States|MUSC Health Florence Medical Center (Site 210-006), 805 Pamplico Highway, Florence, South Carolina, United States|VA TVHS Nashville Campus (Site 074-022), 1310 24th Avenue South, Nashville, Tennessee, United States|Vanderbilt University Medical Center (Site 212-001), 1211 Medical Center Drive, Nashville, Tennessee, United States|Hendrick Medical Center (Site 080-014), 1900 Pine Street, Abilene, Texas, United States|CHRISTUS Spohn Shoreline Hospital (Site 080-001), 600 Elizabeth Street, Corpus Christi, Texas, United States|Parkland Health and Hospital Systems (Site 084-002), 5200 Harry Hines Blvd, Dallas, Texas, United States|UT Southwestern Medical Center (Site 084-001), 1936 Amelia Court, 2nd Floor, Dallas, Texas, United States|Baylor, Scott and White Health (Site 301-003), Baylor University Medical Center, 3500 Gaston Ave., Dallas, Texas, United States|Memorial Hermann Hospital (Site 203-006), 6411 Fannin Street, Houston, Texas, United States|Michael E. DeBakey Veterans Affairs Medical Center (MEDV AMC) (Site 074-006), 2002 Holcombe Blvd., Houston, Texas, United States|Texas Heart Institute (Site 301-017), 6770 Bertner, MC4-266, Houston, Texas, United States|CHRISTUS Good Shepherd Medical Center (Site 080-031), 700 E. Marshall Ave., Longview, Texas, United States|Intermountain Medical Center (Site 211-001), 5121 South Cottonwood Street, Murray, Utah, United States|University of Utah Hospital (Site 211-002), 419 Wakara Way, Suite 207, Salt Lake City, Utah, United States|LDS Hospital (Site 211-004), 8th Ave. C Street, Salt Lake City, Utah, United States|University of Virginia Health Systems (Site 301-021), 1215 Lee Street, Charlottesville, Virginia, United States|Virginia Commonwealth University Health System (Site 210-005), 1250 East Marshall Street, Richmond, Virginia, United States|Carilion Roanoke Memorial Hospital (Site 080-018), 1906 Belleview Avenue, Roanoke, Virginia, United States|Salem VA Medical Center (Site 074-014), 1970 Roanoke Blvd., Salem, Virginia, United States|Harborview Medical Center (Site 208-001), 325 9th Avenue, Seattle, Washington, United States|Swedish Hospital First Hill (Site 208-005), 747 Broadway, Seattle, Washington, United States|University of Washington Medical Center - Montlake (Site 208-006), 1959 NE Pacific Street, Seattle, Washington, United States|West Virginia University (Site 301-023), One Medical Center Drive, Morgantown, West Virginia, United States|Hospital Italiano de Buenos Aires (Site 611-002), Pres. Ttd. Gral. Juan Domingo Perón 4190, Buenos Aires, Argentina|Hospital General de Agudos Dr. JM Ramos Mejia (Site 611-001), Urquiza 609, Ciudad Autonoma de Buenos Aire, Argentina|Centro de Educación Médica e Investigaciones Clinicas "Norberto Quirno" CEMIC (Site 611-021), Av. Cnel. Díaz 2423 á, Ciudad Autonoma de Buenos Aire, Argentina|Aalborg Hospital (Site 625-005), Hobrovej 18, Aalborg, Denmark|Aarhus Universitetshospital, Skejby (Site 625-002), Department of Infectious Diseases, Palle Juul-Hensens Boulevard 99, Aarhus N, Denmark|Righospitalet (Site 625-006), Blegdamsvej 9,, Copenhagen Ø, Denmark|Bispebjerg Hospital (Site 625-013), Bispebjerg Bakke 23, Copenhagen, Denmark|Herlev/Gentofte Hospital (Site 625-012), Medicinsk Afdeling, Herlev Ringvej 75, Herlev, Denmark|Nordsjællands Hospital (Site 625-009), Dyrehavevej 29, Hillerød, Denmark|Hvidovre University Hospital, Department of Infectious Diseases (Site 625-001), Kettegård allé 30, Hvidovre, Denmark|Kolding Sygehus (Site 625-011), Medicinsk Afdeling, Sygehusvej 24, Kolding, Denmark|Odense University Hospital (Site 625-004), Infektionsmedicinsk Forskningsenhed, J.B. Winsløwsgade 4, Odense, Denmark|Zealand University Hospital, Roskilde (Site 625-010), Sygehusvej 10, Roskilde, Denmark|AIDS and Clinical Immunology Research Center (Site 627-201), Infectious Diseases, 16 Al. Kazbegi Avenue, Tbilisi, Georgia|Democritus University of Thrace (Site 635-021), University General Hospital of Alexandroupolis, Dragana, Alexandroupolis, Evros, Greece|Evangelismos COVID-19 Unit, (Site 635-020), 1st Dept. of Pulmonary and Critical Care Medicine, Evangelismos General Hospital, Dept., Ipsilantou 45-47, Athens, Greece|1st Respiratory Medicine Dept., Athens University Medical School (Site 635-015), Athens Hospital for Diseases of the Chest "Sotiria Hospital", 152 Mesogeion Ave., Athens, Greece|3rd Dept. of Medicine, Medical School, NKUA (Site 635-022), Sotiria General Hospital, 152 Mesogeion Ave., Athens, Greece|Attikon University General Hospital (Site 635-009), 4th Dept. of Internal Medicine, Medical School, National and Kapodistrian University of Athens, 1 Rimini St., Haidari, Athens, Greece|Chennai Antiviral Research and Treatment Clinical Research Site (Site 612-402), VHS-IDMC, Voluntary Health Services, Rajiv Gandhi Salai, Taramani, Chennai, Tamil Nadu, India|Medical Centre, Voluntary Health Services (Site 612-402), Rajiv Gandhi Salai, Taramani, Chennai, Tamil Nadu, India|Hospital General Dr. Aurelio Valdivieso (Site 653-004), Calzada Porfirio Díaz No. 400, Oaxaca de Juarez, Oaxaca, Mexico|Hospital General Dr. Manuel Gea González (Site 653-003), Av. Calzada de Tlalpan 4800, Colonia Belisario Domínguez Sec XVI Alcaldía Tlalpan, Mexico City, Mexico|Instituto Nacional de Ciencias Medicas y Nutrición Salvador Zubirán (Site 653-001), Av Vasco de Quiroga #15 Belisario Domínguez Secc XVI, Col. Belisario Domínguez Sección XVI, Alcaldía Tlalpan, Mexico City, Mexico|Instituto Nacional de Enfermedades Respiratorias "Ismael Cosío Villegas" (Site 653-002), Calzada de Tlapan No. 4502, Col. Belisario Domínguez Sección XVI Alcaldía Tlalpan, Mexico City, Mexico|Centro de Investigação e Treino em Saúde da Polana Caniço (CISPOC) (Site 634-701), Instituto Nacional de Saúde (INS), Rua da Costa do Sol, 178, Polana Caniço B, Maputo, Mozambique|Institute of Human Virology Nigeria (IHVN) (Site 612-601), Plot 252, Herbert Macaulay Way, Central Business District, Abuja, Nigeria|Wojewódzki Szpital Zakazny (Site 625-302), Wolska 37, Warsaw, Poland|Tan Tock Seng Hospital (Site 612-201), National Centre for Infectious Diseases (NCID), 11 Jalan Tan Tock Seng, Singapore, Singapore|Hospital Universitari Germans Trias i Pujol (Site 626-003), Infectious Disease Unit, Second Floor, Building Maternal, Road Canyet s/n, Badalona, Barcelona, Spain|Hospital Universitari Arnau de Vilanova (Site 626-035), Av. Alcalde Rovira Roure 80, Lleida, Leida, Spain|Hospital Del Mar (Site 626-025), Paseo Maritimo 25-29, Barcelona, Spain|Hospital Universitari Vall d'Hebron (Site 626-033), Passeig de la Vall d'Hebron 119-129, Barcelona, Spain|Hospital Clínic de Barcelona (Site 626-004), Carrer de Villaroel 170, Barcelona, Spain|Hospital Universitario de Bellvitge (Site 626-034), Carrer de la Feixa Llarga, s/n, Barcelona, Spain|Hospital General Universitario Gregorio Marañón (Site 626-001), Dr. Esquerdo, 46, Madrid, Spain|Hospital Clínico San Carlos (Site 626-017), Enfermedades infecciosas, C/Martin Lagos CN, Madrid, Spain|UCICEC (Clinical Trial Unit) Hospital Universitario La Paz (Site 626-012), Paseo de la Castellana 261, 2a planta Hospital Maternal, Madrid, Spain|University Hospital Zurich (Site 621-201), Department of Infectious Diseases and Hospital Epidemiology, Raemistrasse 100, Zürich, Zurich, Switzerland|MRC/UVRI and LSHTM Uganda Research Unit (Site 634-601), Entebbe Regional Referral Hospital, Entebbe, Uganda|Gulu Regional Referral Hospital (Site 634-603), Laroo Division, PO Box 160, Gulu, Uganda|Makerere University Lung Institute (Site 634-604), New Mulago Hospital Complex, Mulago Hill, Kampala, Uganda|St. Francis Hospital, Nsambya (Site 634-607), Nsambya Road Nsambya Hill, P.O. Box 7146, Kampala, Uganda|Lira Regional Referral Hospital (Site 634-605), Lira, Uganda|Masaka Regional Referral Hospital (Site 634-606), MRC/UVRI and LSHTM Uganda Research Unit, Plot 6 Circle Road, PO Box 556, Masaka, Uganda|Central City Clinical Hospital of Ivano-Frankivsk City Council (Site 627-302), Department of Therapy #1, Hetmana Mazepy str. 114, Ivano-Frankivs'k, Ukraine|Royal Victoria Infirmary (Site 634-007), Queen Victoria Road, Newcastle Upon Tyne, Northumbria, United Kingdom|Royal Free Hospital (Site 634-006), Pond Street, Hampstead, London, United Kingdom|Guy's and St. Thomas' NHS Foundation Trust (Site 634-011), London, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Triple (Participant, Care Provider, Investigator)|Primary Purpose: Treatment
- Enrollment - 10000
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Time from randomization to sustained recovery|All-cause mortality|Composite of time to sustained recovery and mortality|Days alive outside short-term acute care hospital|Pulmonary ordinal outcome|Pulmonary+ ordinal outcome|Incidence of clinical organ failure|Composite of death or serious clinical COVID-19 related events|Composite of cardiovascular events and thromboembolic events|Composite of grade 3 and 4 clinical adverse events, serious adverse events (SAEs) or death|Incidence of infusion reactions|Composite of SAEs or death|Change in SARS-CoV-2 neutralizing antibody levels|Change in overall titers of antibodies|Change in neutralizing antibody levels|Incidence of home use of supplemental oxygen above pre-morbid oxygen use|Incidence of no home use of supplemental oxygen above pre-morbid oxygen use
|
|
NCT04713176
|
Efficacy and Safety of DWJ1248 With Remdesivir in Severe COVID-19 Patients |
Recruiting |
Phase 3 |
Feb/02/2021 |
Jun/30/2021 |
- Alternative id - DW_DWJ1248302
- Interventions - Drug: DWJ1248 with Remdesivir|Drug: Placebo with Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - National Medical Center, Seoul, Korea, Republic of
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 1022
- Age - 19 Years and older (Adult, Older Adult)
- Outcome measures - Rate of mortality or ECMO patients|Recovery days|Desirable of Outcome Ranking (DOOR)|Hospitalization period|Rate of Mortality
|
|
NCT04257656
|
A Trial of Remdesivir in Adults With Severe COVID-19 |
Terminated |
Phase 3 |
Feb/06/2020 |
Apr/10/2020 |
- Alternative id - CAP-China remdesivir 2
- Interventions - Drug: Remdesivir|Drug: Remdesivir placebo
- Study type - Interventional
- Study results - No Results Available
- Locations - Bin Cao, Beijing, Beijing, China
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Quadruple (Participant, Care Provider, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 237
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Time to Clinical Improvement (TTCI) [Censored at Day 28]|Clinical status|Time to Hospital Discharge OR NEWS2 (National Early Warning Score 2) of ≤ 2 maintained for 24 hours.|All cause mortality|Duration (days) of mechanical ventilation|Duration (days) of extracorporeal membrane oxygenation|Duration (days) of supplemental oxygenation|Length of hospital stay (days)|Time to 2019-nCoV RT-PCR negativity in upper and lower respiratory tract specimens|Change (reduction) in 2019-nCoV viral load in upper and lower respiratory tract specimens as assessed by area under viral load curve.|Frequency of serious adverse drug events
|
|
NCT04738045
|
Comparison of Remdesivir Versus Lopinavir/ Ritonavir and Remdesivir Combination in COVID-19 Patients |
Recruiting |
Phase 4 |
Nov/01/2020 |
Apr/01/2021 |
- Alternative id - REC-H-PhBSU-21001
- Interventions - Drug: Remdesivir|Drug: Lopinavir/ Ritonavir and Remdesivir combination
- Study type - Interventional
- Study results - No Results Available
- Locations - Beni-suef University, Banī Suwayf, Egypt
- Study designs - Allocation: Non-Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 90
- Age - 18 Years to 80 Years (Adult, Older Adult)
- Outcome measures - Proportion of cured patients in the interventional group versus the proportion of cured patients in the control group|Monitoring of adverse events.
|
|
NCT04409262
|
A Study to Evaluate the Efficacy and Safety of Remdesivir Plus Tocilizumab Compared With Remdesivir Plus Placebo in Hospitalized Participants With Severe COVID-19 Pneumonia |
Completed |
Phase 3 |
Jun/16/2020 |
Mar/08/2021 |
- Alternative id - WA42511|2020-002275-34
- Interventions - Drug: Remdesivir|Drug: Tocilizumab|Drug: Placebo
- Study type - Interventional
- Study results - Has Results
- Locations - Valleywise Health Medical Center, Phoenix, Arizona, United States|eStudySite - Chula Vista - PPDS, Chula Vista, California, United States|Hoag Hospital Irvine, Irvine, California, United States|Providence St Johns Health Center, Santa Monica, California, United States|Yale University School of Medicine; HIV Clinical Trials Program, New Haven, Connecticut, United States|Medstar Georgetown University Hospital, Washington, District of Columbia, United States|Holy Cross Hospital Inc, Fort Lauderdale, Florida, United States|Larkin Community Hospital Palm Springs Campus (Hialeah), Hialeah, Florida, United States|University of Miami Miller School of Medicine; Clinical Reseach Building, Miami, Florida, United States|Larkin Community Hospital, South Miami, Florida, United States|St Luke's Health System; Rheumatology Research, Boise, Idaho, United States|Advocate Christ Medical Center, Oak Lawn, Illinois, United States|Ochsner Clinic Foundation, New Orleans, Louisiana, United States|University of Maryland, Baltimore, Maryland, United States|Boston Medical Center, Boston, Massachusetts, United States|Baystate Medical Center, Springfield, Massachusetts, United States|Henry Ford Medical Center, Novi, Michigan, United States|St. Michael'S Medical Center, Newark, New Jersey, United States|San Juan Oncology Associates, Farmington, New Mexico, United States|Wyckoff Heights Medical Center, Staten Island, New York, United States|Novant Health Clinical Research, Charlotte, North Carolina, United States|OhioHealth Research Institute, Columbus, Ohio, United States|Providence Saint Vincent's Medical Center, Portland, Oregon, United States|Lehigh Valley Hospital, Allentown, Pennsylvania, United States|Thomas Jefferson University, Philadelphia, Pennsylvania, United States|Medical University of South Carolina, Charleston, South Carolina, United States|The Liver Institute at Methodist Dallas, Arlington, Texas, United States|Baylor Scott and White Medical Center - College Station, College Station, Texas, United States|Baylor University Medical Center, Dallas, Texas, United States|Baylor St. Luke's Medical Center, Houston, Texas, United States|Ben Taub General Hospital - HCHD, Houston, Texas, United States|Houston Methodist Hospital, Houston, Texas, United States|Baylor Scott & White Medical Center - Irving, Irving, Texas, United States|Baylor Scott & White Hospital - Plano, Plano, Texas, United States|Baylor Scott & White Health, Temple, Texas, United States|Intermountain LDS Hospital, Salt Lake City, Utah, United States|The Providence Regional Medical Center Everett, Everett, Washington, United States|West Virginia University Hospital, Morgantown, West Virginia, United States|Santa Casa de Misericordia de Belo Horizonte, Belo Horizonte, MG, Brazil|Hospital Nossa Senhora das Graças; Setor de Pesquisa em Neurologia, Curitiba, PR, Brazil|Instituto de Pesquisa Clinica Evandro Chagas - IPEC FIOCRUZ, Rio de Janeiro, RJ, Brazil|CEMEC - Centro Multidisciplinar de Estudos Clínicos, Sao Bernardo Do Campo, SP, Brazil|Hospital de Base de Sao Jose do Rio Preto, Sao Jose do Rio Preto, SP, Brazil|Instituto de Infectologia Emilio Ribas, Sao Paulo, SP, Brazil|Instituto do Coração - HCFMUSP, Sao Paulo, SP, Brazil|Medsi Clinic, Moscow, Adygeja, Russian Federation|O.M. Filatov City Clinical Hospital #15; Department of Surgery, Moskva, Moskovskaja Oblast, Russian Federation|City Pokrovskaya Hospital, Sankt-peterburg, Sankt Petersburg, Russian Federation|City Clinical Hospital # 52, Moscow, Russian Federation|Hospital Universitario de Bellvitge, Hospitalet de Llobregat, Barcelona, Spain|Hospital Universitario Principe de Asturias; Medicina Interna - Servicio de Enfermedades Infecciosas, Alcala de Henares, Madrid, Spain|Hospital Universitario HM Torrelodones, Torrelodones, Madrid, Spain|Hospital General Universitario de Guadalajara, Guadalajara, Spain|Hospital Universitario Fundacion Jimenez Diaz., Madrid, Spain
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 649
- Age - 12 Years and older (Child, Adult, Older Adult)
- Outcome measures - Time to Hospital Discharge or "Ready for Discharge" up to Day 28|Time to Mechanical Ventilation or Death up to Day 28|Clinical Status as Assessed by the Investigator Using a 7-category Ordinal Scale of Clinical Status on Day 14|Time to Death up to Day 28|Time to Death up to Day 60|Time to Improvement of at Least 2 Categories Relative to Baseline on a 7-category Ordinal Scale of Clinical Status up to Day 28|Clinical Status as Assessed by the Investigator Using a 7-category Ordinal Scale of Clinical Status on Day 7|Clinical Status as Assessed by the Investigator Using a 7-category Ordinal Scale of Clinical Status on Day 21|Clinical Status as Assessed by the Investigator Using a 7-category Ordinal Scale of Clinical Status on Day 28|Clinical Status as Assessed by the Investigator Using a 7-category Ordinal Scale of Clinical Status on Day 60|Proportion of Participants Requiring Initiation of Mechanical Ventilation Post-baseline (Participants Who Did Not Require Mechanical Ventilation at Baseline)|Proportion of Participants Who Are Alive and Free of Respiratory Failure at Day 28 and Day 60 (Participants Requiring Mechanical Ventilation at Baseline)|Duration of Mechanical Ventilation (Participants Requiring Mechanical Ventilation at Baseline) up to Day 28|Difference in Mortality at Days 14, 28, and 60|Time to Recovery up to Day 28|Proportion of Participants Who Are Discharged or "Ready for Discharge" up to Day 28|Proportion of Participants Who Require Initiation of Mechanical Ventilation Post-baseline or Die up to Day 28
|
|
NCT04330690
|
Treatments for COVID-19: Canadian Arm of the SOLIDARITY Trial |
Recruiting |
Phase 2 |
Mar/18/2020 |
May/18/2022 |
- Alternative id - 2114
- Interventions - Drug: Interferon beta-1a|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Peter Lougheed Centre, Calgary, Alberta, Canada|Foothills Medical Centre, Calgary, Alberta, Canada|Rockyview General Hospital, Calgary, Alberta, Canada|South Health Campus, Calgary, Alberta, Canada|Royal Alexandra Hospital, Edmonton, Alberta, Canada|Misericordia Community Hospital, Edmonton, Alberta, Canada|University of Alberta Hopsital, Edmonton, Alberta, Canada|Grey Nuns Community Hospital, Edmonton, Alberta, Canada|Interior Health Royal Inland Hospital, Kamloops, British Columbia, Canada|Lions Gate Hospital, North Vancouver, British Columbia, Canada|Richmond Hospital, Richmond, British Columbia, Canada|Vancouver General Hospital, Vancouver Coastal Health, Univeristy of British Columbia, Vancouver, British Columbia, Canada|St Paul's Hospital, Vancouver, British Columbia, Canada|Island Health - Royal Jubilee Hospital, Victoria, British Columbia, Canada|Island Health - Victoria General Hospital, Victoria, British Columbia, Canada|Island Health - Nanaimo Regional General Hospital, Victoria, British Columbia, Canada|St. Boniface Hospital, Winnipeg, Manitoba, Canada|Health Sciences Centre, Winnipeg, Manitoba, Canada|Grace General Hospital, Winnipeg, Manitoba, Canada|Eastern Regional Health Authority, Saint John's, Newfoundland and Labrador, Canada|Nova Scotia Health Authority, Halifax, Nova Scotia, Canada|Royal Victoria Regional Health Centre, Barrie, Ontario, Canada|William Osler Health System - Brampton Civic Hospital, Brampton, Ontario, Canada|William Osler Health System - Etobicoke General Hospital, Etobicoke, Ontario, Canada|St. Joseph's Healthcare Hamilton, Hamilton, Ontario, Canada|Grand River Hospital, Kitchener, Ontario, Canada|St. Mary's General Hospital, Kitchener, Ontario, Canada|St.Joseph's Health Care, London, Ontario, Canada|University Hospital, London, Ontario, Canada|Victoria Hospital, London, Ontario, Canada|Markham Stouffville Hospital, Markham, Ontario, Canada|Trillium Health Partners -Mississauga Site, Mississauga, Ontario, Canada|Trillium Health Partners-Credit Valley Hospital, Mississauga, Ontario, Canada|The Ottawa Hospital - General Campus, Ottawa, Ontario, Canada|Hôpital Montfort, Ottawa, Ontario, Canada|The Ottawa Hospital - Civic Campus, Ottawa, Ontario, Canada|Queensway Carleton Hospital, Ottawa, Ontario, Canada|Niagara Health, Saint Catharines, Ontario, Canada|Scarborough Health Network - Centenary Hospital, Scarborough, Ontario, Canada|Scarborough Health Network - General Hospital, Scarborough, Ontario, Canada|Scarborough Health Network - Birchmount Hospital, Scarborough, Ontario, Canada|Thunder Bay Regional Health Sciences Centre, Thunder Bay, Ontario, Canada|North York General Hospital, Toronto, Ontario, Canada|Humber River Hospital, Toronto, Ontario, Canada|Michael Garron Hospital, Toronto, Ontario, Canada|Sunnybrook Health Sciences Centre, Toronto, Ontario, Canada|St. Michael's Hospital, Toronto, Ontario, Canada|Mount Sinai Hospital, Toronto, Ontario, Canada|Toronto Western Hospital, Toronto, Ontario, Canada|Unity Health Toronto - St. Joseph's Health Centre, Toronto, Ontario, Canada|CISSS de la Montérégie Centre, Greenfield Park, Quebec, Canada|Centre Hospitalier de l'Université de Montréal (CHUM), Montréal, Quebec, Canada|McGill University Health Centre-Glen Site Royal Victoria Hospital, Montréal, Quebec, Canada|CHU de Québec - Université Laval, Québec, Quebec, Canada|Institut Universitaire de Cardiologie et de Pneumologie de Québec- Université Laval, Québec, Quebec, Canada|Centre Intégré Universitaire de Santé et de Services Sociaux de l'Estrie - Centre Hospitalier Universitaire de Sherbrooke, Sherbrooke, Quebec, Canada|Royal University Hospital, Saskatoon, Saskatchewan, Canada
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 2900
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Efficacy of Interventions as assessed by all-cause mortality|Time to improvement of one category from admission|Subject clinical status|Change in Subject clinical status|Oxygen free days|Incidence of oxygen use|Duration of oxygen use|Incidence of new mechanical ventilation|Duration of mechanical ventilation|Duration of hospitalization|Mortality|Cumulative Incidence of Grade 3 and 4 Adverse Events (AEs) and Serious Adverse Events (SAEs)
|
|
NCT04351724
|
Austrian CoronaVirus Adaptive Clinical Trial (COVID-19) |
Recruiting |
Phase 2|Phase 3 |
Apr/16/2020 |
Mar/31/2022 |
- Alternative id - ACOVACT
- Interventions - Drug: Chloroquine or Hydroxychloroquine|Drug: Lopinavir/Ritonavir|Other: Best standard of care|Drug: Rivaroxaban|Drug: Thromboprophylaxis|Drug: Candesartan|Drug: non-RAS blocking antihypertensives|Drug: Remdesivir|Drug: Asunercept 400mg|Drug: Asunercept 100mg|Drug: Asunercept 25mg|Drug: Pentaglobin
- Study type - Interventional
- Study results - No Results Available
- Locations - Medical University of Innsbruck, Innsbruck, Tirol, Austria|Medical University of Graz, Graz, Austria|Kepler University Hospital, Linz, Austria|Medical University of Vienna, Vienna, Austria|Wilhelminenspital, Vienna, Austria|SMZ Süd Kaiser Franz Josef Spital, Vienna, Austria|KH Hietzing, Vienna, Austria|SMZ Baumgartner Höhe Otto Wagner Spital, Vienna, Austria|SMZ Ost Donauspital, Vienna, Austria
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 500
- Age - 18 Years to 99 Years (Adult, Older Adult)
- Outcome measures - sustained improvement (>48h) of one point on the WHO Scale|Time to improvement on WHO Scale|Mean change in the ranking on an ordinal scale from baseline|time to discharge or a National Early Warning Score (NEWS) ≤2 (maintained for 24h), whichever occurs first|change from baseline in National Early Warning Score (NEWS)|Oxygenation free days|Incidence of new oxygen use during the trial|duration of oxygen use during the trial|Ventilator free days until day 29|Incidence of new mechanical ventilation use during the trial|duration of mechanical ventilation use during the trial|Viral load/viral clearance|Duration of Hospitalization|Mortality|Obesity - mortality|Obesity - duration of hospitalization|Obesity - ICU admission|Obesity - new oxygen use|Drug-drug interactions with lopinavir/ritonavir|Renin Angiotensin System (RAS) fingerprint|SpO2/FiO2 ratio|paO/FiO2 ratio|modified Sequential Organ Failure Assessment|C-reactive protein|Interleukin-6|procalcitonin|IgM Concentrations|IgA Concentrations|differential blood counts
|
|
NCT04693026
|
Efficacy of Ramdicivir and Baricitinib for the Treatment of Severe COVID 19 Patients |
Recruiting |
Phase 3 |
Sep/10/2020 |
Mar/05/2021 |
- Alternative id - M.A.R.M.C.D./2020/2637
- Interventions - Drug: Remdesivir|Drug: Baricitinib|Drug: Tocilizumab
- Study type - Interventional
- Study results - No Results Available
- Locations - M. Abdur Rahim Medical College Hospital, Dinajpur, Bangladesh
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 150
- Age - Child, Adult, Older Adult
- Outcome measures - Time to Clinical Improvement (TTCI)|Mortality Rate|Duration of ICU stay|Duration total hospital stay|Rate of daily Supplemental Oxygen Use|Time to Clinical Failure
|
|
NCT04488081
|
I-SPY COVID-19 TRIAL: An Adaptive Platform Trial for Critically Ill Patients |
Recruiting |
Phase 2 |
Jul/31/2020 |
Nov/01/2022 |
- Alternative id - I-SPY-COVID
- Interventions - Drug: Remdesivir|Drug: Pulmozyme|Drug: IC14|Drug: Celecoxib Famotidine|Drug: Narsoplimab|Drug: Aviptadil Acetate|Drug: Cyclosporine
- Study type - Interventional
- Study results - No Results Available
- Locations - University of Alabama at Birmingham, Birmingham, Alabama, United States|UC Davis Medical Center, Davis, California, United States|Long Beach Memorial Medical Center, Long Beach, California, United States|University of Southern California, Los Angeles, California, United States|Hoag Memorial Hospital Presbyterian, Newport Beach, California, United States|University of California San Francisco (UCSF), San Francisco, California, United States|University of Colorado, Aurora, Colorado, United States|Yale Cancer Center, New Haven, Connecticut, United States|Georgetown University, Washington, District of Columbia, United States|Emory University, Atlanta, Georgia, United States|Northwestern University, Chicago, Illinois, United States|University of Iowa, Iowa City, Iowa, United States|Kalispell Regional Medical Center, Kalispell, Montana, United States|Montefiore Medical Center, Bronx, New York, United States|Columbia University Medical Center, New York, New York, United States|Wake Forest Baptist Comprehensive Cancer Center, Winston-Salem, North Carolina, United States|University of Pennsylvania (U Penn), Philadelphia, Pennsylvania, United States|Main Line Health - Lankenau Medical Center, Wynnewood, Pennsylvania, United States|Sanford Health, Sioux Falls, South Dakota, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 1500
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Identify agents that will result in substantial improvements to the clinical condition of participants with COVID-19|Improvement in disease severity|Health care utilization|Frequency of serious AEs|Mortality
|
|
NCT04647695
|
IFN-beta 1b and Remdesivir for COVID19 |
Recruiting |
Phase 2 |
Nov/20/2020 |
Sep/30/2021 |
- Alternative id - UW 20-535
- Interventions - Drug: Interferon beta-1b|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Queen Mary Hospital, Hong Kong, Hong Kong
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 100
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Clinical improvement|Hospitalisation|NPS viral load|TS viral load|Inflammatory markers|Adverse events|Mortality
|
|
NCT04678739
|
Efficacy and Safety of Remdesivir and Tociluzumab for the Management of Severe COVID-19: A Randomized Controlled Trial |
Completed |
Phase 3 |
Aug/15/2020 |
Feb/10/2021 |
- Alternative id - M.A.R.M.C.D./2020/1985
- Interventions - Drug: Remdesivir|Drug: Tocilizumab
- Study type - Interventional
- Study results - No Results Available
- Locations - Chattogram General Hospital, Chittagong, Bangladesh|Cox's Bazar 250 Bed District Sadar Hospital, Cox's Bazar, Bangladesh|M. Abdur Rahim Medical College Hospital, Dinajpur, Bangladesh|M. Abdur Rahim Medical College Hospital, Dinajpur, Bangladesh
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 205
- Age - 16 Years to 80 Years (Child, Adult, Older Adult)
- Outcome measures - Time to Clinical Improvement (TTCI)|Duration of ICU Stay|Mortality Rate|Time to Recovery|Hospital stay|Rate of daily Supplemental Oxygen Use|Time to Clinical Failure
|
|
NCT04349410
|
The Fleming [FMTVDM] Directed CoVid-19 Treatment Protocol |
Completed |
Phase 2|Phase 3 |
Apr/11/2020 |
Sep/14/2020 |
- Alternative id - FMTVDM2020
- Interventions - Drug: Hydroxychloroquine, Azithromycin|Drug: Hydroxychloroquine, Doxycycline|Drug: Hydroxychloroquine, Clindamycin|Drug: Hydroxychloroquine, Clindamycin, Primaquine - low dose.|Drug: Hydroxychloroquine, Clindamycin, Primaquine - high dose.|Drug: Remdesivir|Drug: Tocilizumab|Drug: Methylprednisolone|Drug: Interferon-Alpha2B|Drug: Losartan|Drug: Convalescent Serum
- Study type - Interventional
- Study results - No Results Available
- Locations - FHHI-OI-Camelot; QME, Los Angeles, California, United States
- Study designs - Allocation: Randomized|Intervention Model: Factorial Assignment|Masking: Single (Investigator)|Primary Purpose: Treatment
- Enrollment - 1800
- Age - Child, Adult, Older Adult
- Outcome measures - Improvement in FMTVDM Measurement with nuclear imaging.|Ventilator status|Survival status
|
|
NCT04492501
|
Investigational Treatments for COVID-19 in Tertiary Care Hospital of Pakistan |
Completed |
Not Applicable |
Apr/01/2020 |
Jul/20/2020 |
- Alternative id - Sultan Mehmood Kamran 3
- Interventions - Procedure: Therapeutic Plasma exchange|Biological: Convalescent Plasma|Drug: Tocilizumab|Drug: Remdesivir|Biological: Mesenchymal stem cell therapy
- Study type - Interventional
- Study results - No Results Available
- Locations - Pak Emirates Military Hospital, Rawalpindi, Punjab, Pakistan
- Study designs - Allocation: Non-Randomized|Intervention Model: Factorial Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 600
- Age - 18 Years to 90 Years (Adult, Older Adult)
- Outcome measures - survival|duration of hospitalization|Time to resolution of cytokine release storm|Time of viral clearance|Complications
|
|
NCT04944082
|
Remdesivir- Ivermectin Combination Therapy in Severe Covid-19 |
Not yet recruiting |
Phase 4 |
Jul/01/2021 |
Dec/31/2021 |
- Alternative id - Remdesivir-Ivermectin
- Interventions - Drug: Ivermectin
- Study type - Interventional
- Study results - No Results Available
- Locations -
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 60
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - 1- Improvement in level of oxygenation|2- Need for ventilator support|3- Length of hospital stay|4- Development of complication|5- Mortality
|
|
NCT04779047
|
Comparative Therapeutic Efficacy and Safety of Different Antiviral and Anti Inflammatory Drugs in COVID-19 Patients. |
Recruiting |
Phase 4 |
Oct/01/2020 |
Apr/05/2021 |
- Alternative id - REC-H-PhBSU-21011
- Interventions - Drug: Remdesivir|Drug: Hydroxychloroquine|Drug: Tocilizumab|Drug: Lopinavir/ Ritonavir|Drug: Ivermectin
- Study type - Interventional
- Study results - No Results Available
- Locations - Beni-suef University, Banī Suwayf, Egypt
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 150
- Age - 18 Years to 88 Years (Adult, Older Adult)
- Outcome measures - Percentage of clinical cure in each arm
|
|
NCT04560231
|
Remdesivir in COVID-19 Lahore General Hospital |
Recruiting |
Early Phase 1 |
Jun/01/2020 |
Nov/30/2020 |
- Alternative id - LGH003
- Interventions - Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Muhammad Irfan Malik, Lahore, Punjab, Pakistan
- Study designs - Allocation: N/A|Intervention Model: Single Group Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 30
- Age - 15 Years to 80 Years (Child, Adult, Older Adult)
- Outcome measures - Clinical response after administration|Clinical response to treatment|Duration of hospitalization|Supplemental Oxygen Requirement from Baseline
|
|
NCT04315948
|
Trial of Treatments for COVID-19 in Hospitalized Adults |
Recruiting |
Phase 3 |
Mar/22/2020 |
Mar/01/2023 |
- Alternative id - C20-15|101015736
- Interventions - Drug: Remdesivir|Drug: Lopinavir/ritonavir|Drug: Interferon Beta-1A|Drug: Hydroxychloroquine|Other: Standard of care|Drug: AZD7442|Other: Placebo
- Study type - Interventional
- Study results - No Results Available
- Locations - Medizinische Universität Innsbruck, Innsbruck, Austria|Kepler Universitätsklinikum Linz, Linz, Austria|Landeskrankenhaus Salzburg Universitätsklinikum der Paracelsus Medizinischen Privatuniversität, Salzburg, Austria|Hôpital Erasme - Cliniques universitaires de Bruxelles, Brussels, Belgium|Hôpital Saint Luc, Brussels, Belgium|CHU Brugmann, Brussels, Belgium|Hôpital La Citadelle, Liège, Belgium|Pôle Hospitalier Jolimont / site de Mons-Warquignies, Mons, Belgium|Fakultní nemocnice u sv. Anny v Brně, Brno, Czechia|Centre Hospitalier Universitaire Amiens-Picardie, Amiens, France|Centre Hospitalier Regional Metz-Thionville, Ars-Laquenexy, France|Centre Hospitalier Régional Universitaire de Besançon, Besançon, France|Centre Hospitalier Universitaire de Bordeaux, Bordeaux, France|CHU APHP Ambroise-Paré, Boulogne-Billancourt, France|Centre Hospitalier Andrée Rosemon, Cayenne, France|CHU Clermont-Ferrand, Clermont-Ferrand, France|Hospices Civil, Colmar, France|APHP - hôpital Henri-Mondor, Créteil, France|Centre Hospitalier Universitaire Dijon-Bourgogne, Dijon, France|Centre Hospitalier Universitaire de Martinique, Fort De France, France|AP-HP Hôpital Bicêtre, Kremlin-Bicêtre, France|Centre Hospitalo-Universitaire de Grenoble, La Tronche, France|Centre hospitalier de Versailles, Le Chesnay, France|Centre Hospitalier Régional Universitaire de Lille, Lille, France|Hospices Civils de Lyon, Lyon, France|Centre Hospitalier de Mont de Marsan, Mont-de-Marsan, France|Centre Hospitalier Universitaire de Montpellier, Montpellier, France|Groupe Hospitalier de la Région de Mulhouse Sud Alsace, Mulhouse, France|Centre Hospitalier Régional et Universitaire de Nancy, Nancy, France|Centre Hospitalier Universitaire de Nantes, Nantes, France|Centre Hospitalo-Universitaire de Nice, Nice, France|CHU Nîmes, Nîmes, France|APHP - Hôpital Lariboisière, Paris, France|APHP - Hôpital Saint Louis, Paris, France|APHP - Hôpital Saint Antoine, Paris, France|APHP - Hôpital Universitaire Pitié Salpêtrière, Paris, France|APHP - Hôpital Cochin, Paris, France|Hôpital Paris Saint-Joseph et Marie Lannelongue, Paris, France|APHP - Hôpital Necker, Paris, France|APHP- Hôpital Européen Georges-Pompidou, Paris, France|APHP - Hôpital Bichat Claude Bernard, Paris, France|APHP - Hôpital Tenon, Paris, France|CHU Poitiers, Poitiers, France|CH Cornouaille, Quimper, France|CHU de Reims, Reims, France|Centre Hospitalier Universitaire de Rennes, Rennes, France|Hopital DELAFONTAINE, Saint-Denis, France|Hôpital d'Instruction des Armées BEGIN, Saint-Mandé, France|Centre Hospitalier Universitaire de Saint Etienne, Saint-Étienne, France|Centre Hospitalier Régional Universitaire de Strasbourg, Strasbourg, France|Centre Hospitalier Universitaire de Toulouse, Toulouse, France|Centre Hospitalier Universitaire de Toulouse, Toulouse, France|Centre Hospitalier de Tourcoing, Tourcoing, France|Centre Hospitalier Universitaire de Tours, Tours, France|CH Bretagne Atlantique, Vannes, France|CH Bretagne Atlantique, Vannes, France|Centre Hospitalier Annecy Genevois, Épagny, France|Evaggelismos General Hospital, Athens, Greece|General University Hospital of Patras, Patras, Greece|hospital Saint James, Dublin, Ireland|Centre Hospitalier Luxembourg, Luxembourg, Luxembourg|Hôpitaux Robert Schuman, Luxembourg, Luxembourg|Akershus Unniversity Hospital, Oslo, Norway|Lovisenberg Diaconal Hospital, Oslo, Norway|Oslo University Hospital, Oslo, Norway|Bienganski Hospital, Łódź, Poland|Hospital de Abrantes (CHMT), Abrantes, Portugal|Hospital de Cascais, Cascais, Portugal|CHULN- Hospital de Santa Maria, Lisboa, Portugal|CHULN- Hospital de Santa Maria, Lisboa, Portugal|Centro Hospitalar Universitário de São João, EPE, Porto, Portugal|Martin University Hospital, Martin, Slovakia
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Triple (Participant, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 2416
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Percentage of subjects reporting each severity rating on a 7-point ordinal scale|Status on an ordinal scale|National Early Warning Score 2 (NEWS-2 score)|Number of oxygenation free days in the first 28 days|Incidence of new oxygen use, non-invasive ventilation or high flow oxygen devices during the trial.|Ventilator free days in the first 28 days|Incidence of new mechanical ventilation use during the trial.|Need for mechanical ventilation or death by Day 15|Hospitalization|Mortality|Occurrence of new hospitalization|Occurrence of confirmed re-infection with SARS-CoV-2|Cumulative incidence of serious adverse events (SAEs)|Cumulative incidence of Grade 1- 2 hypersensitivity- related and infusion related AEs until D29 visit|Cumulative incidence of Grade 3 and 4 adverse events (AEs)|Number of participants with a discontinuation or temporary suspension of study drugs (for any reason)|Cumulative incidence of AEs of Special Interest
|
|
NCT04728880
|
Remdesivir in Adults With Covid-19: Mansoura University Hospital Experience |
Recruiting |
|
Jan/26/2021 |
May/30/2021 |
- Alternative id - R.20.11.1097
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - Mansoura University Hospital, Mansoura, Dakahliya, Egypt
- Study designs - Observational Model: Cohort|Time Perspective: Retrospective
- Enrollment - 300
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Change from baseline in Pulse rate|Change from baseline in respiratory rate|Change from baseline in body core temperature|Change from baseline in blood pressure|Change from baseline in arterial blood gas analyses|Change from baseline in Spo2 / FIO2 ratio|Change from baseline in White blood cell count|Change from baseline in lymphocyte count|Change from baseline in lactate dehydrogenase|Change from baseline in D-dimer|Change from baseline in procalcitonin|Change from baseline in Interleukin-6|Change from baseline in Serum ferretin|Change from baseline in prothrombin time|Change from baseline in serum creatinine|Change from baseline in liver enzyme ALT|Change from baseline in creatinine kinase|Change from baseline in cardiac troponin|Change from baseline in The Sequential Organ Failure Assessment score (SOFA score)|Duration of hospitalization|Supplemental Oxygen Requirement from Baseline|Duration without mechanical ventilation|Mortality
|
|
NCT04647669
|
World Health Organization (WHO) COVID-19 Solidarity Trial for COVID-19 Treatments |
Not yet recruiting |
Phase 3 |
Jun/01/2021 |
Dec/31/2021 |
- Alternative id - SRD/ETH/20
- Interventions - Drug: Remdesivir|Drug: Acalabrutinib|Drug: Interferon beta-1a|Other: Standard of Care
- Study type - Interventional
- Study results - No Results Available
- Locations - Univeristy of the West Indies, Kingston, Jamaica
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 100
- Age - 18 Years to 100 Years (Adult, Older Adult)
- Outcome measures - All cause Mortality|Duration of hospital stay|Time to first receiving ventilation|Time to admission to the intensive care unit
|
|
NCT04727775
|
Antiviral Drugs on the Treatment of SARS-CoV-2 |
Recruiting |
|
Dec/01/2020 |
Dec/25/2021 |
- Alternative id - AMU
- Interventions - Drug: Favipiravir|Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - Aidos Konkaev, Nur-Sultan, Kazakhstan|Semey Medical University, Semey, Kazakhstan
- Study designs - Observational Model: Cohort|Time Perspective: Retrospective
- Enrollment - 150
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Outcome without comlications|Heart rate|SpO2|Blood pressure|Respiratory rate
|
|
NCT04345419
|
Remdesivir Efficacy in Coronavirus Disease |
Completed |
Phase 2|Phase 3 |
Jun/16/2020 |
Dec/01/2020 |
- Alternative id - COVID 19 treatment
- Interventions - Drug: Remdesivir|Other: Standard of care treatment
- Study type - Interventional
- Study results - No Results Available
- Locations - Tanta university hospital, Assuit University, Ainshams University, Tanta, Egypt
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Single (Participant)|Primary Purpose: Treatment
- Enrollment - 200
- Age - Child, Adult, Older Adult
- Outcome measures - Number of patients with improvement or mortality
|
|
NCT04582266
|
PK and Safety of Remdesivir for Treatment of COVID-19 in Pregnant and Non-Pregnant Women in the US |
Active, not recruiting |
|
Feb/12/2021 |
Apr/01/2022 |
- Alternative id - IMPAACT 2032|DAIDS Study ID 38746
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - USC/Los Angeles County Medical Center NICHD CRS (5048), Los Angeles, California, United States|David Geffen School of Medicine at UCLA (CRS 5112), Los Angeles, California, United States|University California, San Diego (CRS 4601), San Diego, California, United States|Childrens Hospital (U. Colorado, Denver) NICHD CRS (5052), Denver, Colorado, United States|University of Florida (5051), Jacksonville, Florida, United States|Univ of Miami Pediatric/Perinatal HIV/AIDS (4201), Miami, Florida, United States|Emory University School of Medicine (CRS 5030), Atlanta, Georgia, United States|Ann & Robert H Lurie Children's Hospital of Chicago (4001), Chicago, Illinois, United States|Rush University Cook County Hospital NICHD CRS (5083), Chicago, Illinois, United States|Lurie Children's Hospital of Chicago (LCH) CRS, Chicago, Illinois, United States|Johns Hopkins University NICHD CRS (5092), Baltimore, Maryland, United States|Boston Medical Center Ped. HIV Program NICHD CRS (5011), Boston, Massachusetts, United States|Bronx-Lebanon Hospital Center (CRS 5114), Bronx, New York, United States|Jacobi Med. Ctr. Bronx NICHD CRS (5013), Bronx, New York, United States|Stony Brook University Medical Center (CRS 5040), Stony Brook, New York, United States|St. Jude Childrens Research Hosp, Memphis (6501), Memphis, Tennessee, United States|Texas Children's Hospital (Site 5128), Houston, Texas, United States|IMPAACT/ Gamma Project/ UPR Pediatric HIV/AIDS Research CRS, San Juan, Puerto Rico|University of Puerto Rico Pediatric HIV/AIDS CRS (6601), San Juan, Puerto Rico
- Study designs - Observational Model: Cohort|Time Perspective: Prospective
- Enrollment - 40
- Age - Child, Adult, Older Adult
- Outcome measures - PK Outcome: Area under the plasma concentration-time curve (AUC) of RDV|PK Outcome: Half-life (t1/2) of RDV|PK Outcome: Trough concentration (Ctrough) of GS-441524|Safety Outcome: Maternal renal adverse event (AE) of any grade|Safety Outcome: Maternal hepatic AE of any grade|Safety Outcome: Maternal hematologic AE of any grade|Safety Outcome: Maternal Grade 3 or higher AE|Safety Outcome: Serious AE|Safety Outcome: Maternal Grade 3 or higher AE assessed as related to RDV by the Clinical Management Committee (CMC)|Safety Outcome: Pregnancy loss|Safety Outcome: Congenital anomalies|Safety Outcome: Preterm birth, defined as < 37 weeks|Safety Outcome: Preterm birth, defined as < 34 weeks|Safety Outcome: Small for gestational age, defined as < 10th percentile|Safety Outcome: Newborn birth weight|Safety Outcome: Newborn length|Safety Outcome: Newborn head circumference|PK Outcome: AUC of RDV|PK Outcome: t1/2 of RDV|PK Outcome: Ctrough of GS-441524|Safety Outcome: Renal AE of any grade|Safety Outcome: Hepatic AE of any grade|Safety Outcome: Hematologic AE of any grade|Safety Outcome: Grade 3 or higher AE|Safety Outcome: Grade 3 or higher AE assessed as related to RDV by the CMC
|
|
NCT04546581
|
Inpatient Treatment of COVID-19 With Anti-Coronavirus Immunoglobulin (ITAC) |
Completed |
Phase 3 |
Oct/08/2020 |
May/21/2021 |
- Alternative id - INSIGHT 013
- Interventions - Biological: Hyperimmune immunoglobulin to SARS-CoV-2 (hIVIG)|Other: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Penrose Hospital, Colorado Springs, Colorado, United States|St. Francis Health Services, Colorado Springs, Colorado, United States|St. Anthony Hospital, Lakewood, Colorado, United States|Saint Anthony North Health Campus, Westminster, Colorado, United States|Washington VA Medical Center, Washington, District of Columbia, United States|Redmond Regional Medical Center, Rome, Georgia, United States|National Institutes of Health Clinical Center, Bethesda, Maryland, United States|University of Massachusetts, Worcester, Massachusetts, United States|Hennepin Healthcare Research Institute/HCMC, Minneapolis, Minnesota, United States|University of Missouri, Columbia, Missouri, United States|Cox Medical Centers, Springfield, Missouri, United States|FirstHealth Moore Regional Hospital, Pinehurst, North Carolina, United States|Ohio Health Research Institute, Columbus, Ohio, United States|Hendrick Medical Center, Abilene, Texas, United States|CHRISTUS Spohn Shoreline Hospital, Corpus Christi, Texas, United States|University of Texas Southwestern Medical Center, Dallas, Texas, United States|CJW Chippenham Medical Center, Richmond, Virginia, United States|Henrico Doctors' Hospital (HCA), Richmond, Virginia, United States|Aarhus Universitetshospital, Skejby, Aarhus, Denmark|Bispebjerg Hospital, Copenhagen, Denmark|CHIP, Department of Infectious Diseases, Section 2100, Copenhagen, Denmark|Herlev-Gentofte Hospital, Hellerup, Denmark|Nordsjællands Hospital, Hillerød, Hillerød, Denmark|Hvidovre University Hospital, Department of Infectious Diseases, Hvidovre, Denmark|Kolding Sygehus, Kolding, Denmark|Odense University Hospital, Odense, Denmark|Democritus University of Thrace, Alexandroupoli, Thrace, Greece|3rd Dept of Medicine, Medical School, NKUA, Athens, Greece|1st Respiratory Medicine Dept, Athens University Medical School, Athens, Greece|Attikon University General Hospital, Athens, Greece|Dept. of Critical Care & Pulmonary Medicine, Evangelismos General Hospital, Athens, Greece|NCGM, Tokyo, Japan|Fujita Health University Hospital, Toyoake, Japan|Institute of Human Virology-Nigeria (IHVN), Abuja, Nigeria|Hospital Universitari Germans Trias i Pujol, Badalona, Barcelona, Spain|Hospital Universitari Vall d'Hebron, Barcelona, Catalonia, Spain|Hospital del Mar, Barcelona, Spain|Hospital Clínic de Barcelona, Barcelona, Spain|Royal Free Hospital, London, United Kingdom
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Quadruple (Participant, Care Provider, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 593
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Ordinal Outcome Scale - Day 7|All-cause mortality through Day 28|Ordinal Outcome Scale|Change in National Early Warning Score (NEWS)|Time to Worsening|Discharge Status|Days Alive Outside the Hospital|Pulmonary-only Components of the Primary Ordinal Outcome|Thrombotic Components of the Primary Ordinal Outcome|Time to recovery|Clinical Organ Dysfunction|Safety and Tolerability - Adverse Events|Safety and Tolerability - Infusion Reactions, Interruptions, or Cessation|Safety and Tolerability - Serious Adverse Events|Safety and Tolerability - Prevalence of Adverse Events|Change in Neutralizing Antibody Level
|
|
NCT04865237
|
SARS-CoV-2 Human Challenge Characterisation Study |
Active, not recruiting |
Not Applicable |
Mar/06/2021 |
Jun/03/2022 |
- Alternative id - 20IC6437
- Interventions - Biological: SARS-CoV-2 Virus|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - hVIVO Services Ltd, QMB Bioenterprise building, London, United Kingdom|Royal Free Foundation Hospital, London, United Kingdom
- Study designs - Allocation: N/A|Intervention Model: Single Group Assignment|Masking: None (Open Label)|Primary Purpose: Diagnostic
- Enrollment - 36
- Age - 18 Years to 30 Years (Adult)
- Outcome measures - To evaluate the safety of wild type SARS-CoV-2 challenge in healthy participants by assessing the occurrence of unsolicited AEs|To evaluate the safety of wild type SARS-CoV-2 challenge in healthy participants by assessing the occurrence of SAEs related to viral challenge|To identify a SARS-Cov-2 inoculum dose that safely induces laboratory confirmed infection in ≥50% of participants (ideally between 50% and 70%).|To further assess SARS-CoV-2 viral infection rates in upper respiratory samples in healthy volunteers, by inoculum dose|To assess the incidence of symptomatic SARS-CoV-2 infection, in healthy volunteers, by inoculum dose|To assess the SARS-CoV-2 viral dynamics (VL-AUC) in upper respiratory samples in healthy volunteers, by inoculum dose|To assess the SARS-CoV-2 induced symptoms, in healthy volunteers, by inoculum dose according to the sum total symptoms diary card score|To assess the incidence of SARS-CoV-2 illness, in healthy volunteers, by inoculum dose|To assess the SARS-CoV-2 viral dynamics (Peak viral load) in upper respiratory samples in healthy volunteers, by inoculum dose|To assess the SARS-CoV-2 viral dynamics (duration) in upper respiratory samples in healthy volunteers, by inoculum dose|To assess the SARS-CoV-2 viral dynamics (Incubation period) in upper respiratory samples in healthy volunteers, by inoculum dose.|To assess the SARS-CoV-2 induced symptoms, in healthy volunteers, by inoculum dose according to area under the curve over time (TSS-AUC) of total clinical symptoms (TSS).|To assess the SARS-CoV-2 induced symptoms, in healthy volunteers, by inoculum dose according to peak symptom diary card scores.|To assess the SARS-CoV-2 induced symptoms, in healthy volunteers, by inoculum dose according to peak daily symptom score.|To assess the SARS-CoV-2 induced symptoms, in healthy volunteers, by inoculum dose according to Number (%) of participants with Grade 2 or higher symptoms
|
|
NCT04843761
|
ACTIV-3b: Therapeutics for Severely Ill Inpatients With COVID-19 |
Recruiting |
Phase 3 |
Apr/20/2021 |
Apr/01/2023 |
- Alternative id - 015 / ACTIV-3b
- Interventions - Biological: Remdesivir|Drug: Remdesivir Placebo|Biological: Aviptadil|Drug: Aviptadil Placebo|Drug: Corticosteroid
- Study type - Interventional
- Study results - No Results Available
- Locations - Banner University Medical Center Tucson (Site 206-004), 1625 N. Campbell Avenue, Tucson, Arizona, United States|UCSF Fresno (Site 203-005), 155 N. Fresno Street, Fresno, California, United States|VA Loma Linda Healthcare System (Site 074-017), 11201 Benton Street, Loma Linda, California, United States|Cedars-Sinai Medical Center (Site 208-002), 8700 Beverly Boulevard, Los Angeles, California, United States|West Los Angeles VA Medical Center (Site 074-001), 11301 Wilshire Blvd., Los Angeles, California, United States|Ronald Reagan UCLA Medical Center (Site 203-002), 757 Westwood Plaza, Los Angeles, California, United States|UCSF Medical Center at Mount Zion (Site 203-007), 1600 Divisadero St., San Francisco, California, United States|UCSF Medical Center (Site 203-001), Moffit-Long Hospital, 505 Parnassus Ave., San Francisco, California, United States|Stanford University Hospital & Clinics (Site 203-003), 300 Pasteur Dr., Stanford, California, United States|University of Colorado Hospital (Site 204-001), 12605 E. 16th Avenue, Aurora, Colorado, United States|Denver Health Medical Center (Site 204-004), 780 Delaware Street, Pavilion B, Denver, Colorado, United States|MedStar Health Research Institute (Site 009-021), 110 Irving St., NW., Washington, District of Columbia, United States|Washington DC VA Medical Center (Site 009-004), 50 Irving Street, NW., Washington, District of Columbia, United States|Emory University (Site 301-008), Bldg. A, Suite 2236, 1365 Clifton Rd., NE., Atlanta, Georgia, United States|Edward Hines VA Hospital (Site 074-012), 5000 S. 5th Ave., Hines, Illinois, United States|Massachusetts General Hospital (Site 202-002), 55 Fruit Street, Boston, Massachusetts, United States|Baystate Medical Center (Site 201-001), Critical Care Research, 759 Chestnut Street, Springfield, Massachusetts, United States|Henry Ford Health System (Site 205-006), 2799 W. Grand Blvd., Detroit, Michigan, United States|Montefiore Medical Center - Moses campus (Site 206-003), 111 E. 210th Street, Bronx, New York, United States|Montefiore Medical Center - Moses Hospital (Site 206-001), 111 E. 210th Street, Bronx, New York, United States|Mount Sinai Medical Center (Site 301-012), Icahn School of Medicine at Mount Sinai, One Gustave L. Levy Place, New York, New York, United States|Columbia University Irving Medical Center (Site 301-027), 177 Fort Washington Ave., New York, New York, United States|Duke University Hospital (Site 301-006), 2301 Erwin Road, Durham, North Carolina, United States|Wake Forest Baptist Health (Site 210-001), Medical Center Blvd, Winston-Salem, North Carolina, United States|University of Cincinnati Medical Center (Site 207-003), 234 Goodman Street, ML 0740, Cincinnati, Ohio, United States|Cleveland Clinic Fairview Hosptial (Site 207-005), 18101 Lorain Ave., Cleveland, Ohio, United States|Cleveland Clinic Foundation (Site 207-001), 9500 Euclid Ave., Cleveland, Ohio, United States|The Ohio State University Wexner Medical Center (Site 207-004), 410 W. 10th Avenue, Columbus, Ohio, United States|Cleveland Clinic Marymount Campus (Site 207-006), 12300 McCracken Road, Garfield Heights, Ohio, United States|Cleveland Clinic Hillcrest Hospital (Site 207-007), 6780 Mayfield Road, Mayfield Heights, Ohio, United States|Oregon Health & Science University (Site 208-003), 3181 SW Sam Jackson Park Rd., Portland, Oregon, United States|Medical University of South Carolina (Site 210-002), 96 Jonathan Lucas St., CSB 214, Charleston, South Carolina, United States|Vanderbilt University Medical Center (Site 212-001), 1211 Medical Center Drive, Nashville, Tennessee, United States|Baylor, Scott and White Health (Site 301-003), Baylor University Medical Center, 3500 Gaston Ave., Dallas, Texas, United States|Houston Methodist Hospital (Site 301-028), 6565 Fannin Street, Houston, Texas, United States|Texas Heart Institute (Site 301-017), 6770 Bertner, MC4-266, Houston, Texas, United States|University of Texas Health Science Center (Site 203-006), 7000 Fannin St., Houston, Texas, United States|Intermountain Medical Center (Site 211-001), 5121 South Cottonwood Street, Murray, Utah, United States|University of Utah Hospital (Site 211-002), 50 North Medical Drive, Salt Lake City, Utah, United States|UVA School of Medicine (Site 210-003), University of Virginia Health System, University Hospital, 1215 Lee St., Charlottesville, Virginia, United States|Harborview Medical Center (Site 208-001), 325 9th Ave., Seattle, Washington, United States|Swedish Medical Center (Site 208-005), 747 Broadway, Seattle, Washington, United States|West Virginia University (Site 301-023), One Medical Center Drive, Morgantown, West Virginia, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Triple (Participant, Care Provider, Investigator)|Primary Purpose: Treatment
- Enrollment - 640
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Recovery, assessed at 90 days|All-cause mortality|Time to death|Composite of time to recovery, days at home off new supplemental oxygen and time to death|Composite of alive, at home and off new supplemental oxygen|Composite of recovered, alive and not recovered, and dead|Time from randomization to recovery|Days alive outside short-term acute care hospital|Incidence of clinical organ failure or serious infections|Composite of death, clinical organ failure or serious infections|Composite of cardiovascular events and thromboembolic events|Composite of grade 3 and 4 clinical adverse events, serious adverse events (SAEs) or death|Incidence of infusion reactions|Percentage of participants for whom infusion was interrupted or stopped prior to completion for any reason|Percentage of participants for whom infusion was interrupted or stopped prior to completion due to adverse event|Composite of hospital readmissions or death|Incidence of no home use of supplemental oxygen above pre-morbid oxygen use|Time to hospital discharge from initial hospitalization|Composite of death or serious clinical COVID-19 related events|Pulmonary ordinal outcome|Composite of SAEs or death|Incidence of home use of supplemental oxygen above pre-morbid oxygen use|In category 4, 5 or 6 at Day 90 vs. in categories 1-3 at Day 90|In category 5 or 6 at Day 90 vs. in categories 1-4 at Day 90
|
|
NCT04847622
|
Study to Evaluate the Clinical Outcomes in Adults With Covid-19 Who Have Been Treated With Remdesivir. |
Recruiting |
|
Feb/01/2021 |
Jun/30/2021 |
- Alternative id - NEAT ID 909REM
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - CHU de Nantes, Nantes, France|Hospital Lariboisiere, Paris, France|Hopital Saint-Louis, Paris, France|Tel Aviv Sourasky Medical Center, Tel Aviv, Israel|Erasmus Medical Center, Rotterdam, Netherlands|Hospital Germans Trias i Pujol, Barcelona, Spain|Hospital Clínic de Barcelona, Barcelona, Spain|Hospital Universitari Vall d'Herbon, Barcelona, Spain|Hospital Clinico Universitario San Carlos, Madrid, Spain|Hospital Universitario 12 de Octubre, Madrid, Spain|Royal Free Hospital, London, United Kingdom|Chelsea & Westminster Hospital, London, United Kingdom
- Study designs - Observational Model: Cohort|Time Perspective: Retrospective
- Enrollment - 450
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - All-cause mortality|Clinical status assessed by a 7-point ordinal scale|Proportion of clinical improvement.|Severity of disease|Time to SpO2|Duration of oxygen therapy|Admission to the Intensive Care Unit|Time on mechanical ventilation/ECMO|Duration of hospitalisation
|
|
NCT04321616
|
The Efficacy of Different Anti-viral Drugs in COVID 19 Infected Patients |
Recruiting |
Phase 2|Phase 3 |
Mar/28/2020 |
Nov/01/2020 |
- Alternative id - 118684
- Interventions - Drug: Hydroxychloroquine|Drug: Remdesivir|Other: (Standard of Care) SoC
- Study type - Interventional
- Study results - No Results Available
- Locations - Andreas Barratt-Due, Oslo, Norway
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 700
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - In-hospital mortality|Occurrence and duration of mechanical ventilation|Occurrence and duration of intensive care unit (ICU) treatment|Duration of hospital admittance|28 Day mortality|Viral clearance as assessed by SARS-CoV-2 PCR in peripheral blood and nasopharyngeal airway speciemen|Occurrence of co-infections|Occurrence of organ dysfunction
|
|
NCT04970719
|
Baricitinib in Hospitalized Covid-19 Patients With Diabetes Mellitus |
Recruiting |
Phase 3 |
Jul/10/2021 |
Dec/01/2021 |
- Alternative id - BADAS-ERC/EC/21/00311
- Interventions - Drug: Baricitinib|Drug: Dexamethasone|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Debidwar Upazila Health Complex, Comilla, Bangladesh|BIRDEM General Hospital, Dhaka, Bangladesh|Mugda Medical College and Hospital, Dhaka, Bangladesh|Kurmitola General Hospital, Dhaka, Bangladesh|Kurigram Adhunik Sadar Hospital, Kurigram, Bangladesh|Rajshahi Medical College & Hospital, Rajshahi, Bangladesh|Dedicated Corona Isolation Hospital (DCIH), Rangpur, Bangladesh
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 382
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Rescue treatment|Death or invasive mechanical ventilation|C-reactive protein (CRP)|lactate dehydrogenase (LDH)|Ferritin|Creatinine|alanine aminotransferase (ALT)|d-dimer concentration|fasting blood glucose (FBS)|hemoglobin|platelets|white blood cell count (WBC)|total lymphocyte count|adverse events (AEs)|serious adverse events (SAEs)|invasive mechanical ventilation|non-invasive ventilation/high flow oxygen|supplemental oxygen|Desirability of Outcome Ranking (DOOR)|Duration of hospitalization|Incidence of discontinuation or temporary suspension of study product administration|Subject 14-day mortality|Subject 28-day mortality|Subject clinical status|The proportion of subjects meeting criteria for each of the 8 ordinal scale categories|The proportion of subjects not meeting criteria for one of the three most severe ordinal scale categories at any time|Time to an improvement of one category from baseline using an ordinal scale|Time to recovery
|
|
NCT04539262
|
Study in Participants With Early Stage Coronavirus Disease 2019 (COVID-19) to Evaluate the Safety, Efficacy, and Pharmacokinetics of Remdesivir Administered by Inhalation |
Completed |
Phase 1|Phase 2 |
Sep/14/2020 |
Mar/22/2021 |
- Alternative id - GS-US-553-9020
- Interventions - Drug: Remdesivir (RDV)|Drug: Placebo
- Study type - Interventional
- Study results - Has Results
- Locations - The Institute for Liver Health, Mesa, Arizona, United States|The Institute for Liver Health, Tucson, Arizona, United States|Franco Felizarta, MD, Bakersfield, California, United States|Aurora FDRC Inc., Costa Mesa, California, United States|Elevated Health, Huntington Beach, California, United States|Cedars-Sinai Medical Center, Los Angeles, California, United States|Western Clinical Research, Placentia, California, United States|UC Davis Health/Medical Center, Sacramento, California, United States|Bradenton Research Center, Inc., Bradenton, Florida, United States|Integrity Clinical Research, LLC, Doral, Florida, United States|Holy Cross Hospital, Fort Lauderdale, Florida, United States|Evolution Clinical Trials, Inc., Hialeah Gardens, Florida, United States|Research in Miami, Inc., Hialeah, Florida, United States|Optimus U Corporation, Miami, Florida, United States|L & C Professional Medical Research Institute, Miami, Florida, United States|Westchester Research Center at Westchester General Hospital, Miami, Florida, United States|MedBio Trials, Miami, Florida, United States|Nuovida Research Center, Corp, Miami, Florida, United States|IMIC Inc, Palmetto Bay, Florida, United States|Triple O Research Institute, PA, West Palm Beach, Florida, United States|Family Care Research, Boise, Idaho, United States|CTU Covid Research Center, New Orleans, Louisiana, United States|STAT Research, Vandalia, Ohio, United States|Inquest Clinical Research, Baytown, Texas, United States|DFW Clinical Research, Dallas, Texas, United States|Baylor Research Institute, Dallas, Texas, United States|PCP for Life-Tidwell, Houston, Texas, United States|Providence Regional Medical Center Everett, Everett, Washington, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Double (Participant, Investigator)|Primary Purpose: Treatment
- Enrollment - 156
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Time-weighted Average Change From Baseline in Nasopharyngeal Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Viral Load Through Day 7|Time-weighted Average Change From Baseline in Oropharyngeal SARS-CoV-2 Viral Load Through Day 7|Time-weighted Average Change From Baseline in Saliva SARS-CoV-2 Viral Load Through Day 7|Percentage of Participants Experiencing Treatment Emergent Adverse Events (TEAEs)|Percentage of Participants With Treatment-Emergent Laboratory Abnormalities as Per Severity Grade|Percentage of Participants With Treatment-Emergent Adverse Events Leading to Study Treatment Discontinuation|Number of Participants With All-Cause Medically Attended Visits (MAVs) or Death by Day 28|Number of Participants With COVID-19 Related MAVs or Death by Day 28|Number of Participants With Hospitalization by Day 28|Pharmacokinetic (PK) Parameter: AUC0-24h of Remdesivir (RDV) and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: AUClast of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: CLss/F of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: t1/2 of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: Vz/F of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: Cmax of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: Tmax of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: Clast of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: Tlast of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: AUCtau of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: λz of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|PK Parameter: Ctau of RDV and Its Metabolites (GS-441524 and GS-704277) in Parts A and B|Change in Nasopharyngeal SARS-CoV-2 Viral Load From Baseline to Day 5|Change in Oropharyngeal SARS-CoV-2 Viral Load From Baseline to Day 5|Change in Saliva SARS-CoV-2 Viral Load From Baseline to Day 5|Change in Nasopharyngeal SARS-CoV-2 Viral Load From Baseline to Day 7|Change in Oropharyngeal SARS-CoV-2 Viral Load From Baseline to Day 7|Change in Saliva SARS-CoV-2 Viral Load From Baseline to Day 7|Change in Nasopharyngeal SARS-CoV-2 Viral Load From Baseline to Day 14 in Parts A and B|Change in Oropharyngeal SARS-CoV-2 Viral Load From Baseline to Day 14 in Parts A and B|Change in Saliva SARS-CoV-2 Viral Load From Baseline to Day 14 in Parts A and B|Time to Negative Nasopharyngeal SARS-CoV-2 Polymerase Chain Reaction (PCR)|Time to Negative Oropharyngeal SARS-CoV-2 Polymerase Chain Reaction (PCR)|Time to Negative Saliva SARS-CoV-2 Polymerase Chain Reaction (PCR)|Time to Alleviation (Mild or Absent) of Baseline COVID-19 Symptoms as Reported on the COVID-19 Adapted InFLUenza Patient-Reported Outcome (FLU-PRO©) Questionnaire in Part C
|
|
NCT04593940
|
Immune Modulators for Treating COVID-19 |
Completed |
Phase 3 |
Oct/15/2020 |
Mar/05/2022 |
- Alternative id - Pro00106301
- Interventions - Drug: Infliximab|Drug: Abatacept|Drug: Remdesivir|Drug: cenicriviroc (closed to enrollment as of 3-Sep-2021)
- Study type - Interventional
- Study results - No Results Available
- Locations - Banner University Medical Center, Phoenix, Arizona, United States|University of Arkansas Medical Sciences, Little Rock, Arkansas, United States|Scripps Clinical Medical Group, La Jolla, California, United States|UCLA - Ronald Reagan Medical Center, Los Angeles, California, United States|Riverside University, Moreno Valley, California, United States|UC Irvine Medical Center, Orange, California, United States|Stanford University Medical Center, Palo Alto, California, United States|UCLA Medical Center- Santa Monica, Santa Monica, California, United States|Medstar Washington Hospital Center, Washington, District of Columbia, United States|University of Florida-Jacksonville, Jacksonville, Florida, United States|University of Illinois at Chicago, Chicago, Illinois, United States|Northwestern University, Chicago, Illinois, United States|Loyola University Medical Center, Maywood, Illinois, United States|University of Iowa, Iowa City, Iowa, United States|University of Kansas, Kansas City, Kansas, United States|University of Kentucky, Lexington, Kentucky, United States|Tulane School of Medicine, New Orleans, Louisiana, United States|University Medical Center New Orleans, New Orleans, Louisiana, United States|Ochsner Medical Center, New Orleans, Louisiana, United States|Anne Arundel Medical Center, Annapolis, Maryland, United States|Johns Hopkins Medical Center, Baltimore, Maryland, United States|Tufts Medical Center, Boston, Massachusetts, United States|Brigham and Women's Hospital, Boston, Massachusetts, United States|Boston Medical Center, Boston, Massachusetts, United States|Beth Israel Deaconess Medical Center, Boston, Massachusetts, United States|U Mass Memorial Medical Center, Worcester, Massachusetts, United States|U Mass University Medical Center, Worcester, Massachusetts, United States|MidMichigan Medical Center- Gratiot, Alma, Michigan, United States|MidMichigan Medical Center - Midland, Midland, Michigan, United States|Mayo Clinic, Rochester, Minnesota, United States|University of Mississippi Medical Center, Jackson, Mississippi, United States|University of Missouri Health Care, Columbia, Missouri, United States|Washington University School of Medicine, Saint Louis, Missouri, United States|Trinitas Hospital, Elizabeth, New Jersey, United States|Hackensack University Medical Center, Hackensack, New Jersey, United States|Rutgers New Jersey Medical School, New Brunswick, New Jersey, United States|NYU Brooklyn, Brooklyn, New York, United States|University at Buffalo, Buffalo, New York, United States|Flushing Hospital Medical Center, Flushing, New York, United States|Jamaica Hospital Medical Center, Jamaica, New York, United States|NYU Long Island, Long Island City, New York, United States|New York University Langone Medical Center, New York, New York, United States|Harlem Hospital Center, New York, New York, United States|Weill Cornell Medicine, New York, New York, United States|St Lawrence Health System, Potsdam, New York, United States|University of Rochester Medical Center-Strong Memorial Hospital, Rochester, New York, United States|University of North Carolina - Chapel Hill, Chapel Hill, North Carolina, United States|Duke University, Durham, North Carolina, United States|Wake Forest University, Winston-Salem, North Carolina, United States|Mercy Saint Vincent Medical Center, Toledo, Ohio, United States|University of Oklahoma Health Sciences Center, Oklahoma City, Oklahoma, United States|Oregon Health and Science University, Portland, Oregon, United States|Temple University Hospital, Philadelphia, Pennsylvania, United States|Reading Hospital Study, Wyomissing, Pennsylvania, United States|Avera McKennan Hospital, Sioux Falls, South Dakota, United States|University of Tennessee Medical Center, Knoxville, Tennessee, United States|Methodist Health System Clinical Research Institute, Dallas, Texas, United States|University of Texas Health Science Center - Houston, Houston, Texas, United States|University of Texas Health Science Center at San Antonio, San Antonio, Texas, United States|Trinity Mother Frances Hospital, Tyler, Texas, United States|University of Texas Health Center at Tyler, Tyler, Texas, United States|University of Utah, Salt Lake City, Utah, United States|Virginia Commonwealth University Medical Center, Richmond, Virginia, United States|University of Washington Medical Center, Seattle, Washington, United States|Providence Medical Research Center, Spokane, Washington, United States|West Virginia University, Morgantown, West Virginia, United States|Gundersen Health System, La Crosse, Wisconsin, United States|Hospital Interzonal Dr Jose Penna Bahia Blanca, Bahía Blanca, Buenos Aires, Argentina|Sanatorio Ramon Cereijo, Caba, Buenos Aires, Argentina|Instituto Medico Platense, La Plata, Buenos Aires, Argentina|Clinica Central S.A., Villa Regina, Rio Negro, Argentina|Hospital Ramos Mejia, Buenos Aires, Argentina|Hospital Rawson, Cordoba, Argentina|Sanatorio Allende, Córdoba, Argentina|Sanatorio Britanico, Rosario, Argentina|Sanatorio Diagnóstico/ Instituto del Buen Aire, Santa Fe, Argentina|Hospital Brasília, Brasília, DF, Brazil|Hospital Felício Rocho, Belo Horizonte, MG, Brazil|Instituto DOR de Ensino e Pesquisa Hospital Glória D'Or, Rio De Janeiro, Rio De Janeiro / RJ, Brazil|Hospital Ernesto Dornelles, Porto Alegre, Rio Grande D Sul /RS, Brazil|Hospital de Clinicas de Porto Alegre HCPA, Porto Alegre, Rio Grande Do Sul / RS, Brazil|Santa Casa de Misericordia de Porto Alegre, Porto Alegre, Rio Grande Do Sul/RS, Brazil|Hospital e Maternidade Celso Pierro - PUC Campinas, Campinas, São Paulo/SP, Brazil|Nuevo Hospital Civil de Guadalajara "Dr. Juan I. Menchaca", Guadalajara, Guadalajara Jalisco, Mexico|Hospital Universitario "Dr. Jose Eleuterio Gonzalez", Nuevo León, Monterrey, Mexico|Hospital Central FAP, Lima, Lima/Lima, Peru|Hospital Regional Lambayeque, Chiclayo, Peru|Hospitala Nacional Hipólito Unánue, Lima, Peru|Hospital Nacional Aezobispo Loayza, Lima, Peru|Hospital de Chancay y Servicios Basicos de Salud, Lima, Peru|Clínica Belén SANNA, Piura, Peru
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Triple (Participant, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 1971
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Number of patients that recovered from COVID-19|Change in number of patients hospitalized on invasive mechanical ventilation|number of patients that improved clinically|Number of patient deaths|Number of patients with decreased supplemental oxygenation needed|Change in number of patients needing non-invasive ventilation/ high flow oxygen|Number of days patients are in the hospital|Number of SAEs and AEs of grade 3 and 4|Number of patients with changes in abnormal WBC counts
|
|
NCT04784559
|
Trial to Determine the Efficacy/Safety of Plitidepsin vs Control in Patients With Moderate COVID-19 Infection |
Recruiting |
Phase 3 |
Jun/04/2021 |
Dec/01/2022 |
- Alternative id - APL-D-003-20|2020-005951-19
- Interventions - Drug: Plitidepsin|Drug: Dexamethasone|Drug: Remdesivir|Drug: Favipiravir
- Study type - Interventional
- Study results - No Results Available
- Locations - Instituto Medico Platense S.A., La Plata, Buenos Aires, Argentina|Clinica Privada Monte Grande S.A, Monte Grande, Buenos Aires, Argentina|Clinica Central S.A., Villa Regina, Rio Negro, Argentina|Sanatorio Parque - Rosario, Rosario, Santa Fe, Argentina|Hospital General Agudos Ignacio, Buenos Aires, Argentina|Hospital Francisco Muñiz, Ciudad autónoma de Buenos Aires, Argentina|Hospital Rawson, Cordoba, Argentina|Hospital São Rafael, Salvador, BA, Brazil|Chronos Pesquisa Clínica, Brasília, DF, Brazil|Hospital Felicio Rocho, Belo Horizonte, MG, Brazil|Santa Casa de Misericordia de Passos, Passos, MG, Brazil|CePCLIN - Centro de Estudos e Pesquisas em Moléstias Infecciosas Ltda, Natal, RN, Brazil|Hospital Moinhos de Vento (HMV), Porto Alegre, RS, Brazil|Hospital São José, Criciúma, SC, Brazil|CEMEC - Centro Multidisciplinar de Estudos Clínicos, São Bernardo Do Campo, Brazil|University Multiprofile Hospital for Active Treatment Sveta Ekaterina EAD, Dimitrovgrad, Bulgaria|Specialized Hospital for Active Treatment of Pneumo-Phthisiatric Diseases - Haskovo, Haskovo, Bulgaria|MHAT "Dr. Nikola Vasiliev" AD, Kyustendil, Bulgaria|Military Medical Academy - MBAL Pleven, Pleven, Bulgaria|"Specialised Hospital for Active Treatment for Pneumophthisiatric Diseases Dr. Dimitar Gramatikov - Ruse" Ltd Department of Pneumology and Phthisiatry, Ruse, Bulgaria|SHATPPD Dr. Dimitar Gramatikov, Ruse Ltd., Ruse, Bulgaria|University First MHAT "St.Yoan Krastitel"-Sofia EAD, Sofia, Bulgaria|University Multiprofile Hospital for Active Treatment ACIBADEM CITY CLINIC TOKUDA HOSPIAL, Sofia, Bulgaria|"MHAT "Sveta Anna"" - Sofia AD, Sofia, Bulgaria|CliniSalud del Sur S.A.S - Centro de Investigación, Envigado, Antioquia, Colombia|Organización Clinica Bonnadona Prevenir S.A.S, Barranquilla, Atlantico, Colombia|Clínica de la Costa Ltda., Barranquilla, Atlántico, Colombia|Sociedad de Cirugía de Bogotá; Hospital de San José, Bogotá, Bogotá D.C., Colombia|Hospital Universitario MEDERI, Bogotá, Bogotá D.C., Colombia|Caja de Compensacion Familiar de Caldas, Manizales, Caldas, Colombia|CH Valence, Valence, Drome, France|Centre Hospitalier Universitaire (CHU) Dijon Bourgogne - Hopital Francois Mitterand, Dijon, France|Nouvel Hôpital Civil Service des maladies infectieuses, Strasbourg, France|Centre Hospitalier Regional et Universitaire de Tours (CHRU Tours) - Hopital Bretonneau, Tours, France|Democritus University Hospital University General Hospital of Alexandroupolis, Alexandroupoli, Greece|Evangelismos Hospital General Hospital of Athens Evangelismos, Intensive Care Unit, Athens, Greece|Sotiria Hospital General Hospital of Chest Diseases of Athens "Sotiria" 3rd Department of Internal Medicine of University of Athens, Athens, Greece|General Hospital of Athens Alexandra, Athens, Greece|General Hospital of Athens "Laiko", University of Athens Agiou, Athens, Greece|Attikon Hospital, Chaïdári, Greece|Tzaneio Hospital General Hospital of Piraeus Tzaneio, Piraeus, Greece|Hospital Cardiologica Aguascalientes, Aguascalientes, Ags, Mexico|Centro Médico ABC, Mexico City, Cdmx, Mexico|Hospital Médica Sur, Mexico City, Cdmx, Mexico|INER, Mexico City, Cdmx, Mexico|Sanatorio Palmore, A.C., Chihuahua, CHH, Mexico|Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán, Mexico City, DIF, Mexico|Centro de Investigación Clínica Chapultepec, Morelia, Michoacan, Mexico|Hospital Español, Ciudad de México, Méx, Mexico|Hospital Angeles (Lomas), Mexico City, Méx, Mexico|Universidad Autonoma de Nuevo Leon - Hospital Universitario "Dr. Jose Eleuterio Gonzalez", Monterrey, NL, Mexico|Hospital Alberto Sabogal Sologuren, Bellavista, Lima, Peru|Hospital de Chancay, Chancay, Lima, Peru|Hospital Nacional Hipolito Unanue (HNHU), El Agustino, Lima, Peru|Universidad Peruana Cayetano Heredia (UPCH) - Hospital Cayetano Heredia (HCH), Lima Cercado, Lima, Peru|HULC - Hospital de Curry Cabral, Lisboa, Portugal|Spitalul Clinic De Boli Infectioase Cluj-Napoca, Sectia HIV/SIDA, Cluj-Napoca, Cluj, Romania|Spitalul Clinic de Boli Infectioase Constanta, Constanţa, Tomis, Romania|Institutul National De Boli Infectioase "Prof. Dr. Matei Bals", Bucharest, Romania|Spitalul Clinic de Boli Infectioase si Tropicale Dr. Victor Babes - Bucharest, Bucharest, Romania|Institutul National De Boli Infectioase "Prof. Dr. Matei Bals", Bucharest, Romania|Spitalul Clinic Universitar de Urgenta Bucuresti, Bucharest, Romania|Spitalul Clinic De Boli Infectioase "Sfanta Parascheva" IASI, Sectia Boli Infectioase III, Iaşi, Romania|Spitalul Judetean de Urgenta 'Sf. Ioan cel Nou' Suceava, Sectia de Boli Infectioase, Suceava, Romania|Netcare Lakeview Hospital, Benoni, Gauteng, South Africa|Tiervlei Trial Centre, Cape Town, Western Cape, South Africa|TASK eden, George, Western Cape, South Africa|Hospital Universitari Germans Trias i Pujol, Badalona, Barcelona, Spain|Hospital Universitari de Bellvitge, Hospitalet de Llobregat, Barcelona, Spain|Hospital Universitario Puerto Real, Puerto Real, Cádiz.Spain, Spain|Hospital Universitario de Jerez de la Frontera, Jerez De La Frontera, Cádiz, Spain|Hospital Universitario HM Montepríncipe, Boadilla Del Monte, Madrid, Spain|Hospital Universitario de Fuenlabrada, Fuenlabrada, Madrid, Spain|Hospital Universitario de Getafe, Getafe, Madrid, Spain|HM Puerta del Sur, Móstoles, Madrid, Spain|Hospital Universitario de Móstoles, Móstoles, Madrid, Spain|Hospital Quirónsalud Madrid, Pozuelo De Alarcón, Madrid, Spain|HM Torrelodones, Torrelodones, Madrid, Spain|Hospital Costa Del Sol, Marbella, Málaga, Spain|Hospital Quirón Marbella, Marbella, Málaga, Spain|Hospital Álvaro Cunqueiro, Vigo, Pontevedra, Spain|Hospital Universitario de Cruces, Barakaldo, Vizcaya, Spain|Hospital General Universitario de Alicante, Alicante, Spain|Complejo Asistencial Universitario de Burgos - Hospital Universitario de Burgos, Burgos, Spain|Universidad de Cadiz (UCA) - Hospital Universitario Puerta del Mar (HUPM), Cadiz, Spain|Hospital Universitario Virgen de las Nieves (HUVN), Granada, Spain|Hospital Clinico San Cecilio, Granada, Spain|Hospital Universitario de Guadalajara, Guadalajara, Spain|Hospital Universitari Arnau de Vilanova de Lleida, Lleida, Spain|H.U. La Princesa, Madrid, Spain|Hospital Gregorio Marañón, Madrid, Spain|Hospital Universitario Moncloa, Madrid, Spain|Hospital Infanta Leonor, Madrid, Spain|Hospital Universitario Ramón y Cajal, Madrid, Spain|Fundacion Jimenez Diaz, Madrid, Spain|Hospital Clínico San Carlos, Madrid, Spain|Hospital 12 Octubre, Madrid, Spain|H. HM Sanchinarro, Madrid, Spain|Hospital de Emergencias Enfermera Isabel Zendal, Madrid, Spain|Complexo Hospitalario de Pontevedra, Pontevedra, Spain|Hospital Universitario de Salamanca, Salamanca, Spain|Instituto de Investigación Sanitaria Valdecilla (IDIVAL), Santander, Spain|Hospital Universitario Virgen del Rocío, Sevilla, Spain|Ege University Medical School, Department of Infectious Diseases and Clinical Microbiology, Bornova, İzmir, Turkey|T.C. Saglik Bakanligi Tepecik Egitim ve Arastirma Hastanesi - Enfeksiyon Hastaliklari ve Klinik Mikrobiyoloji Klinigi, Konak, Izmir, Turkey|Kocaeli Universitesi - Kocaeli Universitesi Tip Fakultesi - Kocaeli Universitesi Arastirma ve Uygulama Hastanesi, İzmit, Kocaeli, Turkey|Hacettepe University, School of Medicine, Ankara, Turkey|Ankara City Hospital, Ankara, Turkey
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 609
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Percentage of patients who achieve complete recovery:|Time to complete recovery|Clinical status assessed by 11-category WHO Clinical Progression Scale:|Proportion of patients with treatment-emergent adverse events (TEAEs)|Proportion of patients with Grade ≥3 TEAEs|Proportion of patients with serious adverse events (SAEs)|Proportion of patients with serious adverse reactions (SARs)|Proportion of patients with adverse events (AEs) of special interest|Number and severity of treatment emergent adverse events as per National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0 criteria (CTCAE v5.0)|Proportion of patients requiring re-admission for COVID-19 signs or symptoms|Duration of oxygen therapy (in days)|Proportion of patients requiring high-flow oxygen|Proportion of patients requiring noninvasive mechanical ventilation|Proportion of patients requiring invasive mechanical ventilation or extracorporeal membrane oxygenation (ECMO)|Proportion of patients requiring admission to Intensive Care Unit (ICU)|Duration of hospitalization in ICU|Proportion of patients receiving subsequent antiviral therapies or immunomodulatory drugs|Proportion of patients with nosocomial infection|Mortality|Change in SARS-CoV-2 viral, as measured by quantitative polymerase chain reaction (qPCR) from samples of oro-nasopharyngeal exudate|Proportion of patients with undetectable SARS-CoV-2 viral load, as measured by qPCR from samples of oro-nasopharyngeal exudate|Change in inflammatory biomarker: C-reactive protein (CRP)|Change in inflammatory biomarker: ferritin|Change in inflammatory biomarker: IL-6, IL-1β, IL-10|Change in inflammatory biomarker: tumour necrosis factor alpha (TNFα)|Proportion of patients with serologic response anti-SARS-CoV-2|Time to therapy intensification (WHO >6 [intubation] or initiation of other antiviral/immunomodulating agent)|Percentage of patients requiring increased oxygen therapy on study
|
|
NCT04570982
|
Clinical Protocol for Convalescent Plasma and Remdesivir Therapy in Nepal |
Recruiting |
|
Jul/30/2020 |
Dec/30/2020 |
- Alternative id - NHRC2020-001
- Interventions - Biological: Convalescent Plasma
- Study type - Observational
- Study results - No Results Available
- Locations - Narayani Hospital, Birgunj, Nepal|Seti Provincial Hospital, Dhangadi, Nepal|BP Koirala Institute of Health Sciences (BPKIHS), Dharān Bāzār, Nepal|Sukraraj Tropical Disease Hospital, Kathmandu, Nepal|TU Teaching Hospital, Kathmandu, Nepal|Bheri Provincial Hospital, Nepalgunj, Nepal
- Study designs - Observational Model: Case-Crossover|Time Perspective: Prospective
- Enrollment - 200
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Availability of convalescent plasma|Amount of Plasma|Demographics of recipients|Co-morbidity of recipient|Donor status|Adverse events of convalescent COVID-19 plasma and Remdesivir Therapy|Hospital and ICU length of stay|Disposition of patients including survival
|
|
NCT04832880
|
Factorial Randomized Trial of Rendesivir and Baricitinib Plus Dexamethasone for COVID-19 (the AMMURAVID Trial) |
Not yet recruiting |
Phase 3 |
Apr/06/2021 |
Dec/01/2022 |
- Alternative id - The AMMURAVID trial|2020-001854-23
- Interventions - Drug: Baricitinib Oral Tablet [Olumiant]|Drug: Remdesivir|Drug: Dexamethasone
- Study type - Interventional
- Study results - No Results Available
- Locations - Ospedali Riuniti delle Marche, Ancona, Italy|Ospedale Parini, Aosta, Italy|Ospedale SS Annunziata -Chieti, Chieti, Italy|Ospedale S Anna, Como, Italy|Ospedale di Ferrara, Ferrara, Italy|Ospedale di Firenze and Empoli, Firenze, Italy|Ospedali Galliera, Genova, Italy|H Goretti, Latina, Italy|Ospedale Manzoni, Lecco, Italy|Ospedale di Legnago, Legnago, Italy|Ospedale di Legnano, Legnano, Italy|ASST Fatebenefratelli-Sacco, Milan, Italy|ASST Santi Paolo e Carlo, Milan, Italy|IRCCS San Raffaele, Milan, Italy|Ospedale di Perugia, Perugia, Italy|Ospedale San Salvatore, Pesaro, Italy|Ospedali di Prato e Pistoia, Prato, Italy|Policlinico Tor Vergata, Roma, Italy|Ospedale Cattinara e Maggiore, Trieste, Italy|Ospedale di Udine, Udine, Italy|Azienda Ospedaliera Integrata -Verona, Verona, Italy
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 4000
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Prevention of very severe respiratory failure or mortality|Prevention of mortality|Prevention of very severe respiratory failure|Incidence of Adeverse Events|Incidence of bacterial/fungal infections|Reduction of the requirements of orotracheal intubation/ECMO|Evolution of the NEWS-2 score|Evolution of the MELD score|Velocity in clinical improvement|Velocity in discharge|Fever disappearance|Changes in periperal blood leukocyte number|Changes in periperal blood neutrophils counts|Changes in periperal blood lymphocytes|Changes in periperal blood platelets|Changes in blood hemoglobin levels|Changes in blood creatinine levels|Changes in blood albumin|Changes in blood bilirubin|Changes in blood LDH|Changes in blood AST|Changes in blood ALT|Changes in blood CK|Changes in blood C-reactive protein|Changes in blood IL-6|Changes in blood protrombine time (INR)|Changes in blood ferritin|Changes in blood troponin T|Changes in blood triglycerides|Changes in blood HDL-colesterol|Changes in blood total colesterol|Changes in blood D-Dimer|Changes in PaO2 at arterial gas analysis|Changes in PaO2/FiO2|Development of late complications
|
|
NCT04365725
|
Multicenter, Retrospective Study of the Effects of Remdesivir in the Treatment of Severe Covid-19 Infections |
Completed |
|
May/05/2020 |
Jun/12/2020 |
- Alternative id - APHP200522
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - Hôpital Cochin, Paris, France
- Study designs - Observational Model: Cohort|Time Perspective: Retrospective
- Enrollment - 84
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Clinical course on Day 15.|Clinical course on Day 3.|Clinical course on Day 8|Clinical course on Day 11.|Clinical course on Day 29.|Duration of treatment|Sepsis-related Organ Failure Assessment score|Duration without mechanical ventilation|Mortality|cumulative incidence of grade 3 and 4 adverse events (AEs).
|
|
NCT05185284
|
Randomized Multicenter Study on the Efficacy and Safety of Favipiravir for Parenteral Administration Compared to Standard of Care in Hospitalized Patients With COVID-19 |
Completed |
Phase 3 |
Aug/11/2021 |
Dec/30/2021 |
- Alternative id - FAV-052021
- Interventions - Drug: Favipiravir|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Regional budgetary health care institution "Ivanovo Clinical Hospital named after Kuvaevs", Ivanovo, Russian Federation|State Budgetary Institution of Healthcare of the City of Moscow "City Clinical Hospital No. 24 of the Department of Healthcare of the City of Moscow", Moscow, Russian Federation|State Clinical Hospital №50, Moscow, Russian Federation|Regional Clinic Hospital of Ryazan, Ryazan', Russian Federation|Medical institute Ogarev Mordovia State university, Saransk, Russian Federation|Smolensk clinical hospital №1, Smolensk, Russian Federation
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 217
- Age - 18 Years to 80 Years (Adult, Older Adult)
- Outcome measures - Rate of Clinical Status Improvement|Time to Clinical Improvement|End of Fever on Days of study|Change in the Level of Lung Damage According to CT|Rate of Viral Elimination|Rate of Transfer to the Intensive Care Unit|Rate of the Use of Non-invasive Lung Ventilation|Rate of the Use of Mechanical Ventilation|Mortality
|
|
NCT04596839
|
Antiviral Activity and Safety of Remdesivir in Bangladeshi Patients With Severe Coronavirus Disease (COVID-19) |
Completed |
Phase 2 |
Sep/04/2020 |
Apr/30/2021 |
- Alternative id - BEX-06001
- Interventions - Drug: Remdesivir|Other: Standard of Care
- Study type - Interventional
- Study results - No Results Available
- Locations - Combined Military Hospital, Dhaka, Bangladesh
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 60
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Duration of hospital stay (Days)|Time to Clinical Improvement (TTCI)|All causes mortality|Duration (days) of mechanical ventilation|Duration (days) of supplemental oxygenation|Time to 2019-nCoV RT-PCR negativity in Nasopharyngeal Swab|Frequency of serious adverse drug events
|
|
NCT05041907
|
Finding Treatments for COVID-19: A Trial of Antiviral Pharmacodynamics in Early Symptomatic COVID-19 (PLATCOV) |
Recruiting |
Phase 2 |
Sep/30/2021 |
Aug/01/2023 |
- Alternative id - VIR21001
- Interventions - Drug: Favipiravir|Drug: Monoclonal antibodies|Drug: Ivermectin|Other: No treatment|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Universidade Federal de Minas Gerais, Minas Gerais, Brazil|Vajira hospital, Bangkok, Thailand|Faculty of Tropical Medicine, Mahidol University, Bangkok, Thailand|Bangplee Hospital, Samut Prakan, Thailand
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 750
- Age - 18 Years to 50 Years (Adult)
- Outcome measures - Rate of viral clearance for repurposed drugs|Rate of viral clearance of positive control (monoclonal antibodies) over time relative to the negative control|Rate of viral clearance for small novel molecule drugs|Viral kinetic levels in early COVID-19 disease|Number of antiviral treatment arms that show a positive signal (>90% probability of >5% acceleration in viral clearance)|Rates of viral clearance by treatment arm, as compared against REGN-COV2 (monoclonal antibody cocktail)
|
|
NCT04853901
|
Remdesivir Efficacy In Management Of COVID-19 Patients |
Completed |
Phase 3 |
Jul/27/2020 |
Mar/01/2021 |
- Alternative id - FMASU P56a/ 2020
- Interventions - Drug: Remdesivir|Drug: Standard of care_1|Drug: Standard of care_2
- Study type - Interventional
- Study results - No Results Available
- Locations - Faculty of Medicine Ain Shams University Research Institute- Clinical Research Center, Cairo, Non-US, Egypt
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 77
- Age - 18 Years to 80 Years (Adult, Older Adult)
- Outcome measures - Evaluation of viral clearance
|
|
NCT04669990
|
Remdesivir and Convalescent Plasma Therapy for Treatment of COVID-19 Infection in Nepal : A Registry Study |
Recruiting |
|
Nov/19/2020 |
Nov/19/2021 |
- Alternative id - 749-2020
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - Narayani Hospital, Birgunj, Nepal|Seti Provincial Hospital, Dhangadi, Nepal|BP Koirala Institute of Health Sciences (BPKIHS), Dharān Bāzār, Nepal|Bheri Provincial Hospital, Nepalgunj, Nepal
- Study designs - Observational Model: Case-Only|Time Perspective: Prospective
- Enrollment - 2000
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Demographics of recipients|Co-morbidity of recipient|Adverse events of convalescent COVID-19 plasma and Remdesivir Therapy|Hospital and ICU length of stay|Disposition of patients including survival
|
|
NCT04302766
|
Expanded Access Remdesivir (RDV; GS-5734™) |
No longer available |
|
Jan/01/1970 |
Jan/01/1970 |
- Alternative id - S-20-01
- Interventions - Drug: Remdesivir
- Study type - Expanded Access:Intermediate-size Population|Treatment IND/Protocol
- Study results - No Results Available
- Locations - Naval Medical Center San Diego, San Diego, California, United States|Naval Hospital Jacksonville, Jacksonville, Florida, United States|Benning Martin Army Community Hospital, Fort Benning, Georgia, United States|Eisenhower Army Medical Center, Fort Gordon, Georgia, United States|Tripler Army Medical Center, Tripler AMC, Hawaii, United States|Blanchfield Army Community Hospital, Fort Campbell North, Kentucky, United States|Walter Reed National Military Medical Center, Bethesda, Maryland, United States|Naval Medical Center Camp Lejeune, Camp Lejeune, North Carolina, United States|Womack Army Medical Center, Fort Bragg, North Carolina, United States|William Beaumont Army Medical Center, El Paso, Texas, United States|Carl R. Darnall Army Medical Center, Fort Hood, Texas, United States|Brooke Army Medical Center, Fort Sam Houston, Texas, United States|Naval Medical Center Portsmouth, Portsmouth, Virginia, United States|Madigan Army Medical Center, Tacoma, Washington, United States|Craig Joint Theater Hospital, Bagrām, Afghanistan|NATO Role 3 Multinational Medical Unit Kandahar Air Field, Kandahar, Afghanistan|EMF Camp Lemonnier, Djibouti, Djibouti|Landstuhl Regional Medical Center, Landstuhl, APO Ae, Germany|US Naval Hospital Guam, Agaña, Guam|Baghdad Diplomatic Support Center, Baghdad, Iraq|US Naval Hospital Okinawa, Okinawa, Japan|US Military Hospital Kuwait 411th Hospital Center, Kuwait, Kuwait
- Study designs -
- Enrollment -
- Age - Child, Adult, Older Adult
- Outcome measures -
|
|
NCT05226533
|
Clinical Trials to Assess Safety and Efficacy of DWRX2003 Combination With Remdesivir in Moderate to Severe COVID-19 Patients. |
Not yet recruiting |
Phase 2 |
Feb/02/2022 |
Jul/11/2022 |
- Alternative id - DW_DWJ1516202
- Interventions - Drug: DWRX2003|Drug: Placebo
- Study type - Interventional
- Study results - No Results Available
- Locations -
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Quadruple (Participant, Care Provider, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 60
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - The proportion of subjects requiring mechanical ventilation and invasive mechanical ventilation or extracorporeal membrane oxygenation (ECMO) till day 14.|Change in WHO clinical progression scale from baseline to day 4, 7, 10, 14, 21 and 28.|Change in NEWS2 from baseline to day 4, 7, 10, 14, 21 and 28.
|
|
NCT04410354
|
Study of Merimepodib in Combination With Remdesivir in Adult Patients With Advanced COVID-19 |
Terminated |
Phase 2 |
Jun/16/2020 |
Dec/01/2020 |
- Alternative id - VC-02-01
- Interventions - Drug: Merimepodib|Drug: Matching Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Mayo Clinic in Arizona, Phoenix, Arizona, United States|Holy Cross Hospital, Fort Lauderdale, Florida, United States|Mayo Clinic in Florida, Jacksonville, Florida, United States|Mayo Clinic, Rochester, Minnesota, United States|Atlantic Health System / Morristown Medical Center, Morristown, New Jersey, United States|Atlantic Health System / Overlook Medical Center, Summit, New Jersey, United States|St. David's South Austin Medical Center, Austin, Texas, United States|St. David's Medical Center, Austin, Texas, United States|HCA Houston Healthcare Medical Center, Houston, Texas, United States|HCA Houston Healthcare Mainland, Texas City, Texas, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Triple (Participant, Care Provider, Investigator)|Primary Purpose: Treatment
- Enrollment - 44
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Number of subjects not hospitalized or, if hospitalized, free of respiratory failure|Adverse Events|National Institute of Allergy and Infectious Disease (NIAID) 8-Point Ordinal Scale|Temperature|Death|Mechanical ventilation|Vasopressor Support|Oxygen Therapy|Cessation of Viral Shedding|Change in Oxygen Saturation/Fraction of Inspired Oxygen
|
|
NCT04694612
|
Efficacy of Favipiravir in Treatment of Mild & Moderate COVID-19 Infection in Nepal |
Recruiting |
Phase 3 |
Jan/01/2021 |
May/31/2021 |
- Alternative id - 683-2020
- Interventions - Drug: Favipiravir|Drug: Placebo|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Armed Police Force Hospital, Kathmandu, Bagmati, Nepal|Charak Memorial Hospital, Pokhara, Gandaki, Nepal
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 676
- Age - 18 Years to 80 Years (Adult, Older Adult)
- Outcome measures - clinical improvements in mild cases|clinical improvements in moderate cases|Clinical deterioration in mild & moderate cases|Radiological improvement in moderate cases|28 days mortality in mold & moderate cases|symptomatic improvement or worsening in mild & moderate cases|compare change in SARS-CoV-2 viral load in nasopharyngeal swab in mild & moderate cases|Length of stay in hospital beyond 10 days in mild & moderate cases
|
|
NCT05222113
|
OBSERVATIONAL STUDY OF COMPARISON EFFECTIVITY BETWEEN REMDESIVIR AND FAVIPIRAVIR IN SURABAYA, INDONESIA |
Completed |
|
Aug/01/2021 |
Sep/25/2021 |
- Alternative id - 148/WM12/KEPK/MHSW
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - Gotong Royong Surabaya Hospital, Surabaya, East Java, Indonesia
- Study designs - Observational Model: Case-Only|Time Perspective: Retrospective
- Enrollment - 87
- Age - 18 Years to 97 Years (Adult, Older Adult)
- Outcome measures - Clinical manifestation
|
|
NCT04854837
|
Safety of Remdesivir Treatment in COVID-19 Patients Requiring Hemodialysis |
Recruiting |
|
Apr/12/2021 |
Jul/01/2022 |
- Alternative id - REM-HD
- Interventions - Drug: Remdesivir
- Study type - Observational
- Study results - No Results Available
- Locations - Semmelweis University - Department of Internal Medicine and Oncology, Budapest, Hungary
- Study designs - Observational Model: Case-Control|Time Perspective: Prospective
- Enrollment - 60
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Adverse event frequency|Significant ASAT elevation|Significant ALAT elevation|Significant ALP elevation|Significant seBi elevation|Frequency of suspected drug-induced injury|QTc prolongation|Arrhythmia occurence
|
|
NCT04610541
|
REMdesivir-HU Clinical Study and Severe Covid-19 Patients |
Active, not recruiting |
Phase 3 |
Oct/12/2020 |
Nov/30/2021 |
- Alternative id - REM-ENY-01
- Interventions - Drug: Remdesivir-HU
- Study type - Interventional
- Study results - No Results Available
- Locations - Department of Pulmonology Semmelweis University, Budapest, Hungary|National Korányi Institute for Pulmonology, Budapest, Hungary|North - Central Buda Center New St. János Hospital, Budapest, Hungary|Institute of Infectology, University of Debrecen, Debrecen, Hungary|1st Department of Medicine, University of Pécs, Pécs, Hungary|Department of Internal Medicine University of Szeged, Szeged, Hungary|First Department of Internal Medicine, University of Szeged, Szeged, Hungary
- Study designs - Allocation: N/A|Intervention Model: Single Group Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 2000
- Age - 12 Years to 100 Years (Child, Adult, Older Adult)
- Outcome measures - To assess the safety and tolerability of REM use in Hungary in the conditionally approved indication (EMA)|The proportion of patients with at least 1 treatment-emergent adverse event|Proportion of patients with treatment-emergent clinical
|
|
NCT05239988
|
Observational Study in COVID-19 Patients Treated With Remdesivir |
Recruiting |
|
Nov/24/2021 |
Jun/30/2022 |
- Alternative id - FADOI.02.2021
- Interventions - Other: no experimental intervention
- Study type - Observational
- Study results - No Results Available
- Locations - Ospedale "Mons. Giovanni Galliano", Acqui Terme, (al), Italy|Ente Ecclesiastico "F. Miulli", Acquaviva delle Fonti, (ba), Italy|Ospedale di Andria, Andria, (bt), Italy|P.O. di Garbagnate, Garbagnate Milanese, (mi), Italy|Ospedale di Melegnano, Melegnano, (mi), Italy|Ospedale Broni-Stradella, Stradella, (pv), Italy|P.O. di Polistena, Polistena, (rc), Italy|AO "Giuseppe Foscati", Avellino, Italy|Ospedale "S. Paolo", Bari, Italy|Ospedale di Bentivoglio, Bologna, Italy|Ospedale Maggiore di Bologna Medicina D, Bologna, Italy|P.O. "SS. Trinità", Cagliari, Italy|Ospedale M. Bufalini di Cesena, Cesena, Italy|Ospedale di Alba e Bra, Cuneo, Italy|Ospedale di Faenza, Faenza, Italy|AOU Careggi, Firenze, Italy|Ospedale "Careggi", Firenze, Italy|P.O di Frattamaggiore, Frattamaggiore, Italy|Ospedale Galliera, Genova, Italy|Ospedale del Mare, Napoli, Italy|Azienda Ospedaliero Universitaria di Novara, Novara, Italy|A.O.U. di Perugia, Perugia, Italy|Ospedale di Rimini - Medicina 1 COVID, Rimini, Italy|Ospedale di Rimini -Sub intensiva COVID, Rimini, Italy|Ospedale "Vannini", Roma, Italy|Ospedale Fatebenefratelli Isola Tiberina, Roma, Italy|PO SS Annunziata AOU Sassari, Sassari, Italy|Ospedale "San Paolo" di Savona, Savona, Italy|Ospedale "Mauriziano", Torino, Italy|Ospedale "Circolo-Macchi", Varese, Italy
- Study designs - Observational Model: Cohort|Time Perspective: Retrospective
- Enrollment - 600
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - characteristics of COVID-19 patients|methods of treatment with Remdesivir and the main outcomes
|
|
NCT04480333
|
Safety, Tolerability and Pharmacokinetics of Inhaled Nanoparticle Formulation of Remdesivir (GS-5734) and NA-831 |
Recruiting |
Phase 1 |
Sep/15/2020 |
Mar/31/2021 |
- Alternative id - NEUROSIVIR
- Interventions - Drug: Drug: NA-831 - 0.10 mg/kg|Drug: Placebo- 0.10 mg/kg|Drug: Drug: NA-831 - 0.20 mg/kg|Drug: Placebo- 0.20 mg/kg|Drug: Drug: GS-5734 - 1.00 mg/kg|Drug: Placebo- 1.00 mg/kg|Drug: Drug: GS-5734 - 2.00 mg/kg|Drug: Placebo- 2.00 mg/kg|Combination Product: Drugs: NA-831 (0.10 mg/kg) plus GS-5734 (1.00 mg/kg)|Combination Product: Placebo 0.10 mg + 1.00 mg/kg|Combination Product: Drugs: NA-831 (0.20 mg/kg) plus GS-5734 (2.00 mg/kg)|Combination Product: Placebo 0.20 mg + 2.00 mg/kg
- Study type - Interventional
- Study results - No Results Available
- Locations - Coronavirus Research Institute, Sunnyvale, California, United States
- Study designs - Allocation: Randomized|Intervention Model: Crossover Assignment|Masking: Triple (Participant, Care Provider, Investigator)|Primary Purpose: Treatment
- Enrollment - 45
- Age - 21 Years to 50 Years (Adult)
- Outcome measures - Proportion of Participants Experiencing any Treatment-Emergent Adverse Events|Proportion of Participants Experiencing any Treatment-Emergent Graded Laboratory Abnormalities|Maximum Concentration (Cmax) - Pharmacokinetic Assessment|Time to Maximum Concentration (Tmax) - Pharmacokinetic Assessment|AUC calculated from time of administration to the last measurable concentration (AUC0-last) - Pharmacokinetic Assessment|Area Under the Curve Extrapolated to Infinity (AUC0-∞)|Half-Life (t1/2) - Pharmacokinetic Assessment|Volume of Distribution (Vd) - Pharmacokinetic Assessment|Clearance [CL] - Pharmacokinetic Assessment
|
|
NCT05024006
|
Public Health Emergency: SOLIDARITY TRIAL Philippines |
Completed |
Not Applicable |
Apr/23/2020 |
Apr/17/2021 |
- Alternative id - SJREB-2020-20
- Interventions - Drug: Remdesivir|Drug: Hydroxychloroquine|Drug: Lopinavir / Ritonavir|Drug: Interferon beta-1a|Drug: Acalabrutinib
- Study type - Interventional
- Study results - No Results Available
- Locations - Baguio General Hospital, Baguio City, Benguet, Philippines|Cebu Doctor's University Hospital, Cebu City, Cebu, Philippines|Perpetual Succor Hospital Cebu, Cebu City, Cebu, Philippines|Vicente Sotto Memorial Medical Center, Cebu City, Cebu, Philippines|Southern Philippines Medical Center, Davao City, Davao, Philippines|West Visayas University Medical Center, Iloilo City, Iloilo, Philippines|Makati Medical Center, Makati City, Metro Manila, Philippines|Chinese General Hospital, Manila, Metro Manila, Philippines|Manila Doctors Hospital, Manila, Metro Manila, Philippines|ManilaMed - Medical Center Philippines, Manila, Metro Manila, Philippines|San Lazaro Hospital, Manila, Metro Manila, Philippines|UP - Philippine General Hospital, Manila, Metro Manila, Philippines|Asian Hospital and Medical Center, Muntinlupa, Metro Manila, Philippines|Research Institute for Tropical Medicine, Muntinlupa, Metro Manila, Philippines|San Juan de Dios Educational Foundation Inc - Hospital, Pasay, Metro Manila, Philippines|The Medical City, Pasig City, Metro Manila, Philippines|Diliman Doctors Hospital, Quezon City, Metro Manila, Philippines|Fe Del Mundo Medical Center, Quezon City, Metro Manila, Philippines|Lung Center of the Philippines, Quezon City, Metro Manila, Philippines|St Luke's Medical Center Quezon City, Quezon City, Metro Manila, Philippines|University of the East Ramon Magsaysay Memorial Medical Center, Quezon City, Metro Manila, Philippines|World Citi Medical Center, Quezon City, Metro Manila, Philippines|Cardinal Santos Medical Center, San Juan, Metro Manila, Philippines|St Luke's Medical Center Global, Taguig, Metro Manila, Philippines|Batangas Medical Center, Batangas, Philippines
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 1314
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - All-cause mortality|Duration of hospital stay|Time to first receiving ventilation
|
|
NCT04252664
|
A Trial of Remdesivir in Adults With Mild and Moderate COVID-19 |
Suspended |
Phase 3 |
Feb/12/2020 |
Apr/27/2020 |
- Alternative id - CAP-China remdesivir 1
- Interventions - Drug: Remdesivir|Drug: Remdesivir placebo
- Study type - Interventional
- Study results - No Results Available
- Locations - Jin Yin-tan hospital, Wuhan, Hubei, China
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Quadruple (Participant, Care Provider, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 308
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Time to Clinical recoveryTime to Clinical Recovery (TTCR)|All cause mortality|Frequency of respiratory progression|Time to defervescence (in those with fever at enrolment)|Time to cough reported as mild or absent (in those with cough at enrolment rated severe or moderate)|Time to dyspnea reported as mild or absent (on a scale of severe, moderate, mild absent, in those with dyspnoea at enrolment rated as severe or moderate,)|Frequency of requirement for supplemental oxygen or non-invasive ventilation|Time to 2019-nCoV RT-PCR negative in upper respiratory tract specimen|Change (reduction) in 2019-nCoV viral load in upper respiratory tract specimen as assessed by area under viral load curve.|Frequency of requirement for mechanical ventilation|Frequency of serious adverse events
|
|
NCT04575064
|
An International Randomized Trial of Additional Treatments for COVID-19 in Hospitalized Patients Who Are All Receiving the Local Standard of Care - WHO-SOLIDARITY-GERMANY |
Active, not recruiting |
Phase 2|Phase 3 |
Jun/29/2020 |
Nov/01/2022 |
- Alternative id - WHO-SOLIDARITY-GERMANY|2020-001549-38
- Interventions - Other: Standard of Care (SoC)|Drug: Remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Gesundheit Nord gGmbH, Bremen, Germany|Universitätsklinikum Gießen, Gießen, Germany|Medizinische Hochschule Hannover (MHH), Hannover, Germany|Technische Universität München (TUM), München, Germany
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: None (Open Label)|Primary Purpose: Treatment
- Enrollment - 400
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Improvement of clinical status on the ordinal 7-point severity-scale at day 15.|Time to an improvement of one category from admission using the 7-point ordinal scale|Mortality: 28 days mortality; in-house mortality|Hospital stay: Duration of hospital stay due to COVID-19|Oxygen: Need of, time to first receiving and duration of oxygen|Intensive care: Need of, time to first receiving and duration of intensive care|Mechanical ventilation: Need of, time to first receiving and duration of mechanical ventilation|ECMO: Need of, time to first receiving and duration for extracorporeal membrane oxygenation|Superinfections, assessed with pathogen testing|Kidney failure|Myocardial failure|Multiple organ failure
|
|
NCT04391309
|
COVID-19 and Anti-CD14 Treatment Trial |
Recruiting |
Phase 2 |
Apr/12/2021 |
Feb/01/2022 |
- Alternative id - DAIT COVID-19-003|NIAID CRMS ID#: 38756|UM1AI109565
- Interventions - Biological: anti-CD14|Other: Placebo|Drug: remdesivir
- Study type - Interventional
- Study results - No Results Available
- Locations - Harbor - UCLA Medical Center, Torrance, California, United States|University of Miami Medical Center, Miami Beach, Florida, United States|Sarasota Memorial Health Care System, Sarasota, Florida, United States|Emory University Medical Center, Atlanta, Georgia, United States|Ochsner Health, New Orleans, Louisiana, United States|University of Maryland Medical Center, Baltimore, Maryland, United States|Baystate Medical Center, Springfield, Massachusetts, United States|Cleveland Clinic, Cleveland, Ohio, United States|Thomas Jefferson University Hospital, Philadelphia, Pennsylvania, United States|Virginia Mason Medical Center, Seattle, Washington, United States|Harborview Medical Center, Seattle, Washington, United States|Swedish Medical Center, Seattle, Washington, United States|University of Washington Medical Center-Montlake, Seattle, Washington, United States|University of Washington Medical Center, Seattle, Washington, United States
- Study designs - Allocation: Randomized|Intervention Model: Parallel Assignment|Masking: Quadruple (Participant, Care Provider, Investigator, Outcomes Assessor)|Primary Purpose: Treatment
- Enrollment - 300
- Age - 18 Years and older (Adult, Older Adult)
- Outcome measures - Time to Clinical Recovery|Days Alive and Free of Any Episodes of Acute Respiratory Failure through Day 28|Mean Change in the Ordinal Scale (Range 1 [Best] to 8 [Worst])|Ordinal Scale Value on Day 14 (Range 1 [Best] to 8 [Worst])|All-Cause Mortality through Days 28 and 60|Proportion of Participants Alive and Free of Any Episode of Acute Respiratory Failure through Day 28|Days Alive and Free of Invasive Mechanical Ventilation through Day 28|Proportion of Participants Alive and Free of Invasive Mechanical Ventilation through Day 28|Proportion of Participants Alive and Discharged from the Hospital through Day 28|Proportion of Participants who Begin Corticosteroid Therapy for Worsening COVID-19 Illness after Randomization|Serious Adverse Events (SAEs)|Adverse Events (AEs)|Safety of IC14 as Measured by Change from Baseline in ALT and AST Liver Function Tests|Safety of IC14 as Measured by Change from Baseline in Liver Function Measured by Total Bilirubin|Safety of IC14 as Measured by Change from Baseline in Serum Creatinine|Safety of IC14 as Measured by Change from Baseline in Hemoglobin|Safety of IC14 as Measured by Change from Baseline in White Blood Cell Count|Safety of IC14 as Measured by Change from Baseline in Differential White Blood Count|Safety of IC14 as Measured by Change from Baseline in Platelet Count|Safety of IC14 as Measured by Change from Baseline in Prothrombin Time
|