Cyclosporine
A cyclic peptide immunosuppressant.
General information
Cyclosporine is a cyclic non-ribosomal peptide immunosuppressant. It is used in organ transplant patients and patients with various inflammatory conditions (ChEBI).
Cyclosporine on DrugBank
Cyclosporine on PubChem
Cyclosporine on Wikipedia
Synonyms
Cyclosporin A; Cyclosporine A; Ciclosporin
Marketed as
AQUA-STASIS; CEQUA; CYCLO-DERM; CYCLOSPORINE; GENGRAF; HYDRO-STASIS; NEORAL; RESTASIS; SANDIMMUNE; SANGCYA; VERKAZIA
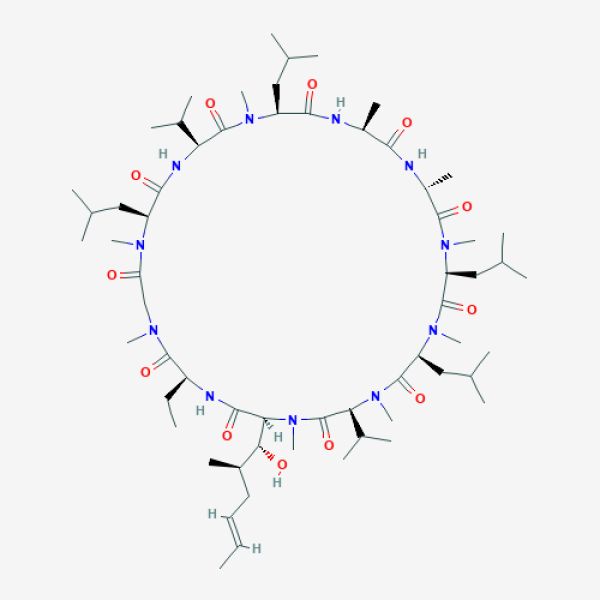
CC[C@H]1C(=O)N(CC(=O)N([C@H](C(=O)N[C@H](C(=O)N([C@H](C(=O)N[C@H](C(=O)N[C@@H](C(=O)N([C@H](C(=O)N([C@H](C(=O)N([C@H](C(=O)N([C@H](C(=O)N1)[C@@H]([C@H](C)C/C=C/C)O)C)C(C)C)C)CC(C)C)C)CC(C)C)C)C)C)CC(C)C)C)C(C)C)CC(C)C)C)C
Supporting references
| Link | Tested on | Impact factor | Notes | Publication date |
|---|---|---|---|---|
|
Discovering drugs to treat coronavirus disease 2019 (COVID-19).
|
in silico | Feb/22/2020 | ||
|
The Host Cell ViroCheckpoint: Identification and Pharmacologic Targeting of Novel Mechanistic Determinants of Coronavirus-Mediated Hijacked Cell States
Preprint |
in silico | one of the top drugs and compounds identified by ViroTreat |
May/17/2020 | |
|
Comparative analysis of antiviral efficacy of FDA-approved drugs against SARS-CoV-2 in human lung cells: Nafamostat is the most potent antiviral drug candidate
Preprint |
Calu-3 human airway epithelial cells | lower IC50 value in Calu-3 cells than VERO E6 cells |
May/12/2020 | |
|
Clinical characteristics and outcomes among hospitalized adults with severe COVID-19 admitted to a tertiary medical center and receiving antiviral, antimalarials, glucocorticoids, or immunomodulation with tocilizumab or cyclosporine...
IL-6 Small molecule Peptide Antibody Cohort study |
Patients | Associated with a significant decrease in mortality. Sample size: 253 + 354 control. Dosage: <5 mg/kg/day for 7-10 days (up to 21 days in severe cases); total cumulative dose >300 mg. |
Oct/15/2020 | |
|
Cyclosporine A plus low‐dose steroid treatment in COVID‐19 improves clinical outcomes in patients with moderate to severe disease. A pilot study
Small molecule Peptide Cohort study |
Patients | 6.87 | The subgroups of severe COVID-19 patients and patients with progressing pneumonia treated with cyclosporine had significantly better outcome compared to control. Lower mortality was observed in the whole treated cohort but especially in moderate to severe patients. Sample size: 105 (cyclosporine with steroids) + 104 control (steroids). Dosage: 1-2 mg/kg daily in two doses for 7 days. Endpoint: Time to clinical improvement until hospital discharge or death (primary). |
Dec/03/2020 |
|
Drug repurposing for COVID-19 using machine learning and mechanistic models of signal transduction circuits related to SARS-CoV-2 infection
Protein factor Small molecule Antibody In silico |
in silico (machine learning) | 13.49 | Considered by the authors to be among the most relevant drugs identified in a machine-learning algorithm-based screening of compounds which considers causal protein-protein interactions, known drug targets, and specific signalling circuits in <a href= |
Dec/11/2020 |
|
Drug repurposing screens reveal cell-type-specific entry pathways and FDA-approved drugs active against SARS-Cov-2
Small molecule In vitro Screening |
Huh7.5 cells; Calu-3 cells; primary normal human bronchial epithelial cells; iPSC-derived AT2 cells; SARS-CoV-2 strain USA WA1/2020 | 8.11 | Inhibited SARS-CoV-2 replication in Huh7.5 cells and also in Calu-3 cells with an SI of >3. |
Mar/23/2021 |
AI-suggested references
Clinical trials
| ID | Title | Status | Phase | Start date | Completion date |
|---|---|---|---|---|---|
| NCT04540926 | Cyclosporine A Plus Low-steroid Treatment in COVID-19 Pneumonia | Not yet recruiting | Phase 1|Phase 2 | Sep/01/2020 | Sep/30/2020 |
|
|||||
| NCT04492891 | Cyclosporine For The Treatment Of COVID-19(+) | Recruiting | Phase 2 | Nov/23/2020 | Nov/23/2025 |
|
|||||
| NCT04488081 | I-SPY COVID-19 TRIAL: An Adaptive Platform Trial for Critically Ill Patients | Recruiting | Phase 2 | Jul/31/2020 | Nov/01/2022 |
|
|||||
| NCT04392531 | Clinical Trial to Assess Efficacy of cYclosporine Plus Standard of Care in Hospitalized Patients With COVID19 | Completed | Phase 4 | Apr/16/2020 | Mar/31/2021 |
|
|||||
| NCT04412785 | Cyclosporine in Patients With Moderate COVID-19 | Completed | Phase 1 | Jun/30/2020 | Oct/25/2021 |
|
|||||
| NCT04979884 | Safety and Effectiveness of Cyclosporin in the Management of COVID19 ARDS Patients in Alexandria University Hospital | Not yet recruiting | Phase 3 | Aug/01/2021 | Apr/01/2022 |
|
|||||
