Boceprevir
A tripeptide hepatitis C virus inhibitor.
General information
Boceprevir is a synthetic tripeptide inhibitor of hepatitis C virus (HCV) genotype 1. It prevents NS3/NS4A protease-mediated viral polyprotein maturation and thereby inhibits viral replication (NCIt).
Boceprevir on DrugBank
Boceprevir on PubChem
Boceprevir on Wikipedia
Synonyms
Victrelis
Marketed as
VICTRELIS
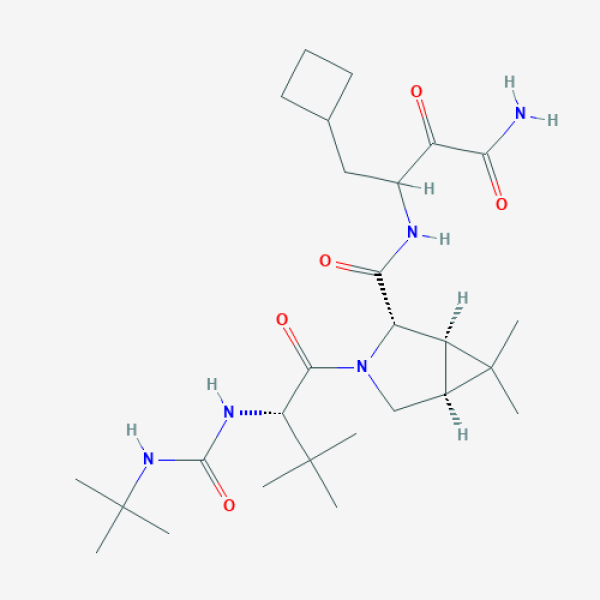
CC1([C@@H]2[C@H]1[C@H](N(C2)C(=O)[C@H](C(C)(C)C)NC(=O)NC(C)(C)C)C(=O)NC(CC3CCC3)C(=O)C(=O)N)C
Supporting references
| Link | Tested on | Impact factor | Notes | Publication date |
|---|---|---|---|---|
|
Predicting commercially available antiviral drugs that may act on the novel coronavirus (2019-nCoV), Wuhan, China through a drug-target interaction deep learning model
Preprint In silico |
in silico | Feb/02/2020 | ||
|
Boceprevir, GC-376, and calpain inhibitors II, XII inhibit SARS-CoV-2 viral replication by targeting the viral main protease
3CLpro Crystallization Small molecule Enzyme assay In vitro |
in vitro enzyme assay; crystallization; Vero 76 cells; SARS-CoV-2 strain USA-WA1/2020 | 20.51 | Inhibited SARS-CoV-2 3C-like protease with an IC50 of ca. 4.13 μM in vitro. Inhibited SARS-CoV-2 infection in primary CPE assay with an EC50 of ca. 1.31 μM and an SI of >76.3. |
Jun/15/2020 |
|
A drug repurposing screen identifies hepatitis C antivirals as inhibitors of the SARS-CoV2 main protease
3CLpro Enzyme assay In vitro In silico Screening |
in silico; in vitro enzyme assay | 2.74 | Inhibited the SARS-CoV-2 3C-like protease in vitro with IC50 of ca. 0.95 μM. |
Feb/01/2021 |
|
Both Boceprevir and GC376 efficaciously inhibit SARS-CoV-2 by targeting its main protease
3CLpro Small molecule Enzyme assay In vitro |
Vero cells; enzyme assay | 12.12 | Inhibit SARS-CoV-2 3C-like protease in vitro via catalytically active site binding. |
Sep/04/2020 |
|
Potential clinical drugs as covalent inhibitors of the priming proteases of the spike protein of SARS-CoV-2
TMPRSS2 Cathepsin B Cathepsin L Small molecule In silico |
in silico | 6.02 | Predicted to inhibit the host TMPRSS2 protease and thereby inhibit the SAS-CoV-2 entry. |
Aug/26/2020 |
|
Malleability of the SARS-CoV-2 3CL Mpro Active-Site Cavity Facilitates Binding of Clinical Antivirals
3CLpro Small molecule Enzyme assay In vitro |
in vitro enzyme assay; X-ray crystallography | 4.86 | When co-crystalized with the SARS-CoV-2 3C-like protease (3CLpro), the drug was observed to interact with and structurally modify the enzyme's active site. The drug also inhibited 3CLpro in vitro. |
Nov/08/2020 |
|
Identification of 14 Known Drugs as Inhibitors of the Main Protease of SARS-CoV-2
3CLpro Small molecule Enzyme assay In vitro |
in vitro enzyme assay | 3.75 | Inhibited the SARS-CoV-2 3C-like protease in vitro with IC50 of 5.4 μM. |
Oct/25/2020 |
|
Identification of existing pharmaceuticals and herbal medicines as inhibitors of SARS-CoV-2 infection
3CLpro PapainLpro Enzyme assay Animal model In vitro In silico |
in silico; in vitro enzyme assay; Vero E6 cells; golden Syrian hamsters; SARS-CoV-2 clinical isolate hCoV-19/Taiwan/4/2020 | 9.41 | Inhibited (not potently) SARS-CoV-2 infection in Vero E6 cells. Inhibited SARS-CoV-2 3C-like protease in vitro. |
Jan/15/2021 |
|
Systematic Search for SARS-CoV-2 Main Protease Inhibitors for Drug Repurposing: Ethacrynic Acid as a Potential Drug
3CLpro Enzyme assay In vitro In silico |
in silico; in vitro enzyme assay | 3.82 | Predicted to inhibit the SARS-CoV-2 3C-like protease. |
Jan/13/2021 |
|
Identification of 3-chymotrypsin like protease (3CLPro) inhibitors as potential anti-SARS-CoV-2 agents
3CLpro Small molecule Enzyme assay In vitro In silico |
in silico; in vitro enzyme assay | Partialy inhibits (IC50 of 31.4 µM; 45% inhibition at 50 µM) the SARS-CoV-2 3C-like protease in vitro. |
Jan/20/2021 | |
|
Development of a Cell-Based Luciferase Complementation Assay for Identification of SARS-CoV-2 3CLpro Inhibitors
3CLpro Small molecule Enzyme assay In vitro |
in vitro enzyme assay | 3.82 | The compound inhibited the SARS-CoV-2 3C-like protease in vitro with EC50 of 38.6 μM and CC50 of >100 μM. |
Jan/24/2021 |
|
Dual inhibition of SARS-CoV-2 and human rhinovirus with protease inhibitors in clinical development
3CLpro Cathepsin L Small molecule Enzyme assay In vitro |
in vitro enzyme assay | 4.10 | The compound inhibited the SARS-CoV-2 3C-like protease with IC50 of 15.6 μM and human cathepsin L with IC50 of 2.3 μM in vitro. The cathepsin L inhibition was relatively weak compared to some other inhibitors. |
Jan/27/2021 |
|
Boceprevir, Calpain Inhibitors II and XII, and GC-376 Have Broad-Spectrum Antiviral Activity against Coronaviruses
Biophysical assay Cathepsin L Enzyme assay In vitro Mechanism |
in vitro enzyme assay; in vitro biophysical assay; Vero cells; Caco-2 cells; (HIV-1) SARS-CoV-2 Spike pseudovirus | 4.61 | The compound inhibited SARS-CoV-2 replication in Caco-2 cells with an EC50 of ca. 2.97 μM and low cytotoxicity (CC50 >100 μM). |
Mar/01/2021 |
